04.06.15
Data from a study comparing Titan Spine’s proprietary surface technology to polyether ether ketone (PEEK) have been published in the March 15 issue of Spine. The data demonstrate that Titan’s line of Endoskeleton interbody devices promote osteoblastic differentiation and enhanced bone-forming environment compared to devices made from PEEK. Specifically, the data show that fibrous tissue formation around PEEK implants may be due to the creation of an inflammatory environment.
“These results indicate that Titan’s surface reduces production of inflammatory mediators and increases production of anti-inflammatory mediators compared to PEEK, thus creating an enhanced environment for bone growth and fusion,” said Barbara Boyan, Ph.D., Dean of the School of Engineering at Virginia Commonwealth University, and lead author of the study. “Fibrous tissue formation around PEEK spinal implants is due to several factors including increased inflammatory cytokines and decreased cell viability. These data add to the growing body of medical knowledge supporting the use of titanium implants featuring a complex roughened topography at the macro-micro-nano levels that induce healing on the cellular level where it is critical for early bone formation.”
This study is yet another example in a rapidly growing body of evidence that PEEK is a poor material for promoting bone growth,” said Peter Ullrich, M.D., former surgeon and CEO of Titan Spine. “Until recently, PEEK was thought to be inert at best. We now understand that is not the case. Dr. Boyan’s research demonstrates that PEEK is actually inhibitory to bone forming cells, called osteoblasts, through the upregulation of pro-inflammatory markers. This leads to fibrous tissue formation, rather than bone formation, as the body attempts to protect itself from PEEK through encapsulation. We were pleased when the IMAST program committee recognized the importance of these data by granting it with the Whitecloud Award. Titan will continue to be at the forefront of scientifically engineering superior interbody fusion devices and promoting titanium as the preferred material for bone growth and fusion.”
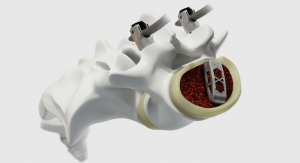
The full line of Endoskeleton devices features Titan Spine’s proprietary implant surface technology, consisting of a combination of roughened topographies at the macro, micro, and cellular levels. This combination of surface topographies is designed to create an optimal host-bone response and actively participate in the fusion process by promoting the upregulation of osteogenic and angiogenic factors necessary for bone growth, encouraging natural production of bone morphogenetic proteins, downregulating inflammatory factors, and creating the potential for a faster and more robust fusion.
Mequon, Wis.-based Titan Spine is a medical device surface technology company focused on developing spinal interbody fusion implants.
“These results indicate that Titan’s surface reduces production of inflammatory mediators and increases production of anti-inflammatory mediators compared to PEEK, thus creating an enhanced environment for bone growth and fusion,” said Barbara Boyan, Ph.D., Dean of the School of Engineering at Virginia Commonwealth University, and lead author of the study. “Fibrous tissue formation around PEEK spinal implants is due to several factors including increased inflammatory cytokines and decreased cell viability. These data add to the growing body of medical knowledge supporting the use of titanium implants featuring a complex roughened topography at the macro-micro-nano levels that induce healing on the cellular level where it is critical for early bone formation.”
This study is yet another example in a rapidly growing body of evidence that PEEK is a poor material for promoting bone growth,” said Peter Ullrich, M.D., former surgeon and CEO of Titan Spine. “Until recently, PEEK was thought to be inert at best. We now understand that is not the case. Dr. Boyan’s research demonstrates that PEEK is actually inhibitory to bone forming cells, called osteoblasts, through the upregulation of pro-inflammatory markers. This leads to fibrous tissue formation, rather than bone formation, as the body attempts to protect itself from PEEK through encapsulation. We were pleased when the IMAST program committee recognized the importance of these data by granting it with the Whitecloud Award. Titan will continue to be at the forefront of scientifically engineering superior interbody fusion devices and promoting titanium as the preferred material for bone growth and fusion.”
The full line of Endoskeleton devices features Titan Spine’s proprietary implant surface technology, consisting of a combination of roughened topographies at the macro, micro, and cellular levels. This combination of surface topographies is designed to create an optimal host-bone response and actively participate in the fusion process by promoting the upregulation of osteogenic and angiogenic factors necessary for bone growth, encouraging natural production of bone morphogenetic proteins, downregulating inflammatory factors, and creating the potential for a faster and more robust fusion.
Mequon, Wis.-based Titan Spine is a medical device surface technology company focused on developing spinal interbody fusion implants.