05.06.15
Dublin, Ireland-based Medtronic plc has earned 510(k) clearance from the U.S. Food and Drug Administration for its Zevo anterior cervical plate system. This system is now commercially available for the treatment of cervical degenerative disc disease, trauma, tumors, deformity, pseudoarthrosis, and/or failed previous fusions.
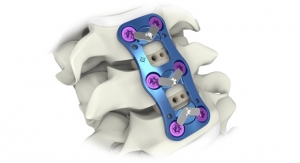
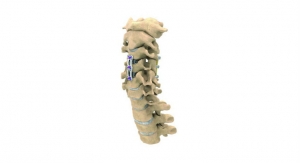
The Zevo system is Medtronic’s latest technology for anterior cervical discectomy with fusion (ACDF) procedures. It features shorter plating options coupled with hyper-screw angulations, allowing the physician to select the smallest possible plate for the patient’s individual surgical needs while securing the plate as far as possible from the adjacent disc. Literature shows this technique may lower the incidence of adjacent level ossification development (ALOD) or bone growth next to the treated level. The Zevo anterior cervical plate and bone screw components are intended for anterior interbody screw fixation from C2-T1.
“Versatility is important in ACDF procedures, and the Zevo anterior cervical plate system was designed to give surgeons more options,” said Doug King, president of the Spinal business and senior vice president of Medtronic. “This system represents Medtronic’s commitment to responding to surgeon needs, and innovating in ways that improve patient treatment and care.”
Additionally, the Zevo system features lower-profile plates with minimal thickness (1.9 mm and 2.1 mm) while increasing the stability, the company claims.
“Improved, thinner plates are important in ACDF procedures,” said Richard Hynes, M.D., spine surgeon at The B.A.C.K. Center in Melbourne, Fla. “Zevo embodies these characteristics, with the added benefits of hyper screw angulations, which can be directed away from the spinal cord.”
The Zevo system is Medtronic’s latest technology for anterior cervical discectomy with fusion (ACDF) procedures. It features shorter plating options coupled with hyper-screw angulations, allowing the physician to select the smallest possible plate for the patient’s individual surgical needs while securing the plate as far as possible from the adjacent disc. Literature shows this technique may lower the incidence of adjacent level ossification development (ALOD) or bone growth next to the treated level. The Zevo anterior cervical plate and bone screw components are intended for anterior interbody screw fixation from C2-T1.
“Versatility is important in ACDF procedures, and the Zevo anterior cervical plate system was designed to give surgeons more options,” said Doug King, president of the Spinal business and senior vice president of Medtronic. “This system represents Medtronic’s commitment to responding to surgeon needs, and innovating in ways that improve patient treatment and care.”
Additionally, the Zevo system features lower-profile plates with minimal thickness (1.9 mm and 2.1 mm) while increasing the stability, the company claims.
“Improved, thinner plates are important in ACDF procedures,” said Richard Hynes, M.D., spine surgeon at The B.A.C.K. Center in Melbourne, Fla. “Zevo embodies these characteristics, with the added benefits of hyper screw angulations, which can be directed away from the spinal cord.”