09.24.15
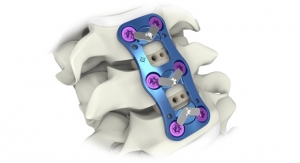
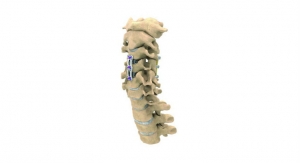
DeGen Medical Inc. has received U.S. Food and Drug Administration clearance for its first cervical spine implant—Hyper-C anterior cervical plate system. The Hyper-C system is an anterior intervertebral fixation device that is designed to aid in stabilizing the cervical spine.
DeGen Medical is a Florence, S.C.-based spinal device development company.
“Clinical research has shown that greater than 5-millimeter (mm) distance—of the cervical plate—from adjacent discs equates to less adjacent level degeneration post-operatively. The small over-hang of Hyper-C plate is an evidence based approach to attain this outcome which may be extremely beneficial for patients in decreasing adjacent level disease,” said Florence-based orthopedic surgeon Rakesh Chokshi, M.D., based on references from clinical articles, though not confirmed with clinical trials.
According to the company, the Hyper-C will be available in the fourth quarter of this year.
“The super-short Hyper-C plate allows for hyper-angulation of bone screws in convergent or divergent orientation,” said W.S. Edwards Jr., M.D., a spinal surgeon also based in Florence. “This allows more intraoperative flexibility for primary and revision surgery with existing hardware.”
The Hyper-C anterior cervical plate system offers plates in levels 1 thru 5 in addition to multiple lengths; bone screws in 4 mm, 4.25 mm, and 4.6 mm diameters; and fixed or variable bone screw choices as well as self-drilling or self-tapping option.
DeGen Medical is a Florence, S.C.-based spinal device development company.
“Clinical research has shown that greater than 5-millimeter (mm) distance—of the cervical plate—from adjacent discs equates to less adjacent level degeneration post-operatively. The small over-hang of Hyper-C plate is an evidence based approach to attain this outcome which may be extremely beneficial for patients in decreasing adjacent level disease,” said Florence-based orthopedic surgeon Rakesh Chokshi, M.D., based on references from clinical articles, though not confirmed with clinical trials.
According to the company, the Hyper-C will be available in the fourth quarter of this year.
“The super-short Hyper-C plate allows for hyper-angulation of bone screws in convergent or divergent orientation,” said W.S. Edwards Jr., M.D., a spinal surgeon also based in Florence. “This allows more intraoperative flexibility for primary and revision surgery with existing hardware.”
The Hyper-C anterior cervical plate system offers plates in levels 1 thru 5 in addition to multiple lengths; bone screws in 4 mm, 4.25 mm, and 4.6 mm diameters; and fixed or variable bone screw choices as well as self-drilling or self-tapping option.