Greatbatch Inc.12.02.15
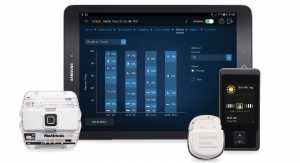
Frisco, Texas-based Greatbatch Inc. has earned approval from the U.S. Food and Drug Administration (FDA) for its Algovita spinal cord stimulation (SCS) system to treat chronic intractable pain of the trunk and/or limbs.
“We are excited to receive PMA approval from the FDA for the Algovita SCS system,” said Thomas J. Hook, president and CEO of Greatbatch. “Algovita’s approval not only supports the proposed Nuvectra spin-off, but the system brings to the U.S. market a robust and user-friendly device for patients suffering from chronic pain of the trunk and/or limbs.”
According to the compny, the Algovita SCS system is the first FDA-approved product developed by Greatbatch’s QiG Group subsidiary. As previously disclosed, Greatbatch is in the process of finalizing the proposed spin-off of QiG Group LLC, which will be renamed Nuvectra Corporation when the process is completed.
Since its inception in 2008, QiG Group has developed a broad-based neurostimulation technology platform. Initially, the company will be focused on the commercialization of the Algovita SCS system, used to treat a variety of neuropathic pain conditions. The Algovita SCS system received CE mark for commercialization in Europe last year, and is currently in limited release in Europe.
The proposed Nuvectra spin-off is expected to be completed in the first quarter of 2016, subject to the Form 10 registration with the U.S. Securities and Exchange Commission statement becoming effective. Form 10 is a general statement of securities.
“We are excited to receive PMA approval from the FDA for the Algovita SCS system,” said Thomas J. Hook, president and CEO of Greatbatch. “Algovita’s approval not only supports the proposed Nuvectra spin-off, but the system brings to the U.S. market a robust and user-friendly device for patients suffering from chronic pain of the trunk and/or limbs.”
According to the compny, the Algovita SCS system is the first FDA-approved product developed by Greatbatch’s QiG Group subsidiary. As previously disclosed, Greatbatch is in the process of finalizing the proposed spin-off of QiG Group LLC, which will be renamed Nuvectra Corporation when the process is completed.
Since its inception in 2008, QiG Group has developed a broad-based neurostimulation technology platform. Initially, the company will be focused on the commercialization of the Algovita SCS system, used to treat a variety of neuropathic pain conditions. The Algovita SCS system received CE mark for commercialization in Europe last year, and is currently in limited release in Europe.
The proposed Nuvectra spin-off is expected to be completed in the first quarter of 2016, subject to the Form 10 registration with the U.S. Securities and Exchange Commission statement becoming effective. Form 10 is a general statement of securities.