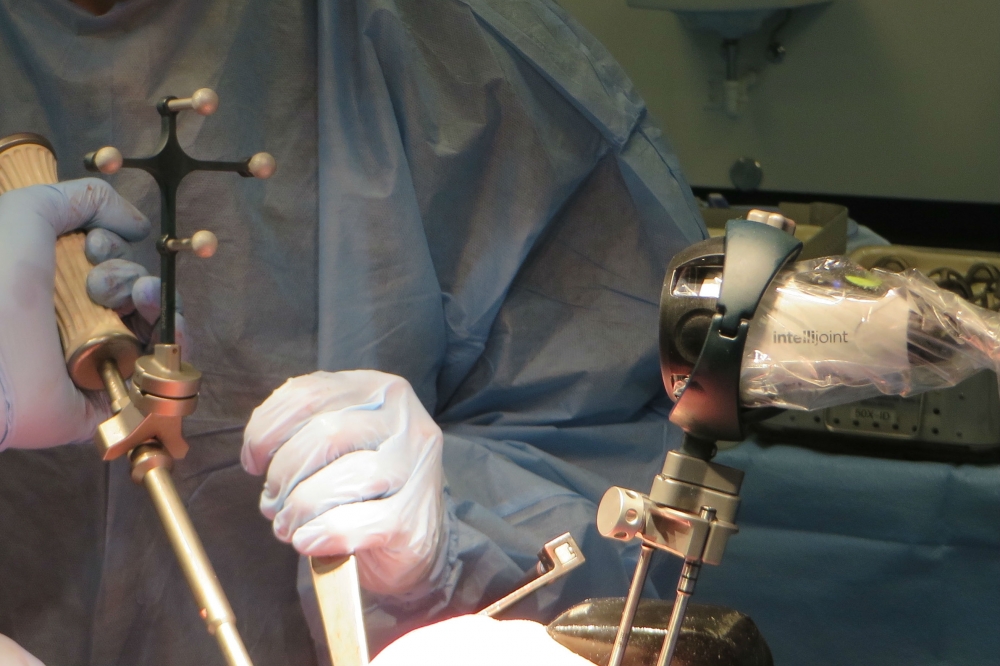
“With the previous generation of the product, our use showed improved accuracy and precision of implant selection without a significant expense or impact on OR time,” said Dr. Wayne Paprosky, professor and orthopedic surgeon at Rush Medical Center in Chicago, Ill. “The new generation of intellijoint HIP provides surgeons with additional critical implant alignment measurements including cup inclination and anteversion, which will be a potential game changer of how hip surgery is performed in the U.S.”
intellijoint HIP can assist in the prevention of recurrent instability, hip dislocation and leg length discrepancy and may expedite decision-making and improve accuracy in achieving preoperative planned targets, ultimately improving patient satisfaction and healthcare economics, according to company executives.
“Intellijoint was founded with the vision of developing and commercializing a technology that is capable of providing real-time 3-D surgical measurement data, without adding significant time, expense or complexity to surgery,” said Armen Bakirtzian, CEO and co-founder of Intellijoint Surgical. “Now, with FDA clearance of intellijoint HIP, we’ve realized this vision and are ready to bring its benefits to the largest market in the world.”
Intellijoint’s core technology represents the latest in miniature 3-D surgical measurement within the sterile field. intellijoint HIP is the first of many products to benefit from this new novel core technology, officials said. It is safe, fast, accurate, and precise. intellijoint HIP‘s compatibility with the majority of implant vendors, paired with its cost-effectiveness, opens accessibility of computer-assisted surgery to all orthopedic surgeons.
Intellijoint Surgical is a Waterloo, Ontario-based company. Founded in 2010, it develops and commercializes miniaturized surgical smart tools that enhance surgeon accuracy, increase hospital economic efficiencies, and improve patient outcomes.