Business Wire07.12.16
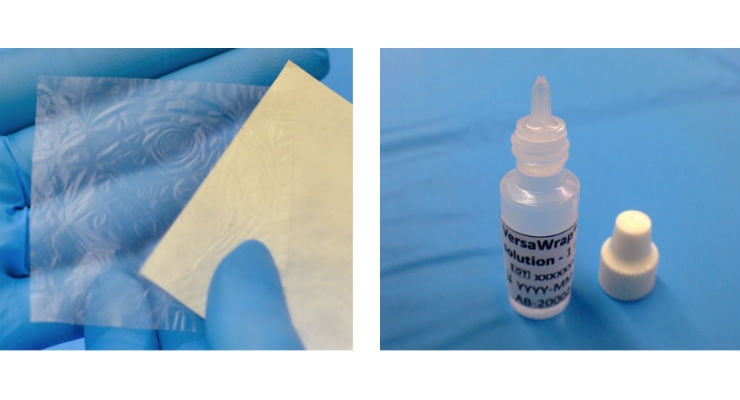
Alafair Biosciences Inc., an emerging medical device company, announced today (11 July) that it received 510(k) clearance from the FDA to market and sell its tendon product, VersaWrap Tendon Protector, in the US. VersaWrap Tendon Protector is an ultrathin, sutureless, bioresorbable hydrogel that protects tendon gliding by separating injured tendon from surrounding tissues. Alafair will formally introduce the VersaWrap Tendon Protector to surgeons at the American Society for Surgery of the Hand meeting in Austin, TX.
The properties of VersaWrap Tendon Protector include an ultrathin profile allowing for application between tight tissues, a sutureless application eliminating the need for sutures or tissue glue, and device bioresorption following tendon healing.
“This ultrathin product gives surgeons the ability to protect tendon gliding in confined areas such as those found in upper extremity,” said Brent Egeland, MD, a hand and plastic surgeon, “and the VersaWrap application procedure is sutureless, which makes it quick and easy to use.”
The VersaWrap Tendon Protector is the first product in the VersaWrap family of products to receive clearance and is indicated for the management and protection of tendon injuries in which there has been no substantial loss of tendon tissue.
The properties of VersaWrap Tendon Protector include an ultrathin profile allowing for application between tight tissues, a sutureless application eliminating the need for sutures or tissue glue, and device bioresorption following tendon healing.
“This ultrathin product gives surgeons the ability to protect tendon gliding in confined areas such as those found in upper extremity,” said Brent Egeland, MD, a hand and plastic surgeon, “and the VersaWrap application procedure is sutureless, which makes it quick and easy to use.”
The VersaWrap Tendon Protector is the first product in the VersaWrap family of products to receive clearance and is indicated for the management and protection of tendon injuries in which there has been no substantial loss of tendon tissue.