Business Wire04.19.17
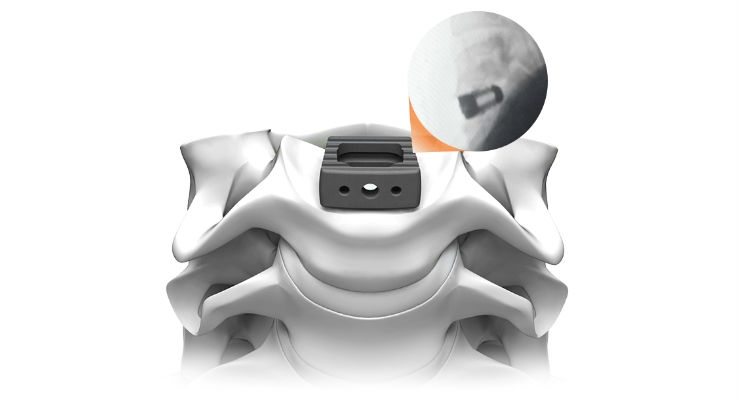
Life Spine, a medical device company that designs, develops, manufactures, and markets products for the surgical treatment of spinal disorders, announced that the U.S. Food and Drug Administration (FDA) has given 510(k) market clearance to the PLATEAU-C Ti Cervical Spacer System.
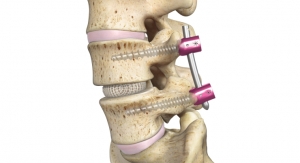
PLATEAU-C Ti joins Life Spine’s extensive portfolio of 62 product lines cleared by the FDA. The system offers Life Spine’s proprietary titanium surface technology, OSSEO-LOC, which is an innovative surface technology that helps create an environment for potential bone growth. Additionally, in comparison to its PEEK counter-part, PLATEAU-C Ti offers additional graft windows for increased visibility in-situ and bone graft containment.
“The new PLATEAU-C Ti with OSSEO-LOC surface technology makes a great addition to an already robust interbody portfolio which includes PEEK, titanium, expandable, and stand-alone technologies. As one of the fastest-growing companies in our space it is important to our surgeons and patients that we continue to advance the cutting edge of technology,” said Mariusz Knap, vice president of marketing for Life Spine.
PLATEAU-C Ti entered a Full Market Release on April 3, 2017.
PLATEAU-C Ti joins Life Spine’s extensive portfolio of 62 product lines cleared by the FDA. The system offers Life Spine’s proprietary titanium surface technology, OSSEO-LOC, which is an innovative surface technology that helps create an environment for potential bone growth. Additionally, in comparison to its PEEK counter-part, PLATEAU-C Ti offers additional graft windows for increased visibility in-situ and bone graft containment.
“The new PLATEAU-C Ti with OSSEO-LOC surface technology makes a great addition to an already robust interbody portfolio which includes PEEK, titanium, expandable, and stand-alone technologies. As one of the fastest-growing companies in our space it is important to our surgeons and patients that we continue to advance the cutting edge of technology,” said Mariusz Knap, vice president of marketing for Life Spine.
PLATEAU-C Ti entered a Full Market Release on April 3, 2017.