Business Wire06.19.17
The Medicrea Group, pioneering the convergence of healthcare IT and next-generation, outcome-centered device design and manufacturing with UNiD ASI technology, announced that it has received FDA 510(k) Clearance and performed first surgeries with PASS TULIP top-loading fixation including world-first patient-specific hybrid constructs.
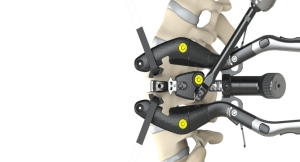
Top-loading fixation is the global standard for posterior spinal instrumentation. Medicrea’s PASS TULIP provides a procedurally-integrated solution for surgeons to benefit from the company’s UNiD ASI technology, a scientific, data-driven model for personalized spine care. The PASS TULIP components are fully compatible with Medicrea’s PASS LP to provide a unique hybrid approach in complex indications. The first-ever patient-specific hybrid spine surgery was successfully performed by Dr. Frank Schwab, Chief of Spine, at the Hospital for Special Surgery in New York.
“By using two compatible implant designs in a hybrid application, I am able to personalize a patient’s operation in a whole new way. The increased flexibility and precision help me to best achieve the patient’s optimal spinal alignment targeted by Medicrea’s patient-specific implants,” stated Dr. Schwab, who uses UNiD ASI technology to strategically plan cases supported by the UNiD LAB team. “Having these additional tools to achieve my surgical strategy translates to better alignment post-operatively and that’s clearly been shown to correlate with patient satisfaction and with long-term benefits of surgery.”
Denys Sournac, president and CEO, stated, “We are pleased to extend our distinctive Lifetime Warranty covering UNiD Rod constructs to include associated PASS TULIP components.” Sournac continued, “The addition of PASS TULIP to our comprehensive implant range will open new doors for Medicrea to gain market share for our UNiD ASI technology by lowering the barrier to entry for the large number of surgeons trained on top-loading instrumentation.”
PASS TULIP components are differentiated from traditional top-loading implants by a thoughtful proprietary design that allows a single implant to perform multiple clinical functions through a simple one-step maneuver. The anticipated result is increased surgical efficiency in implant placement as well as a reduction in the inventory required for a case and associated processing costs.
Top-loading fixation is the global standard for posterior spinal instrumentation. Medicrea’s PASS TULIP provides a procedurally-integrated solution for surgeons to benefit from the company’s UNiD ASI technology, a scientific, data-driven model for personalized spine care. The PASS TULIP components are fully compatible with Medicrea’s PASS LP to provide a unique hybrid approach in complex indications. The first-ever patient-specific hybrid spine surgery was successfully performed by Dr. Frank Schwab, Chief of Spine, at the Hospital for Special Surgery in New York.
“By using two compatible implant designs in a hybrid application, I am able to personalize a patient’s operation in a whole new way. The increased flexibility and precision help me to best achieve the patient’s optimal spinal alignment targeted by Medicrea’s patient-specific implants,” stated Dr. Schwab, who uses UNiD ASI technology to strategically plan cases supported by the UNiD LAB team. “Having these additional tools to achieve my surgical strategy translates to better alignment post-operatively and that’s clearly been shown to correlate with patient satisfaction and with long-term benefits of surgery.”
Denys Sournac, president and CEO, stated, “We are pleased to extend our distinctive Lifetime Warranty covering UNiD Rod constructs to include associated PASS TULIP components.” Sournac continued, “The addition of PASS TULIP to our comprehensive implant range will open new doors for Medicrea to gain market share for our UNiD ASI technology by lowering the barrier to entry for the large number of surgeons trained on top-loading instrumentation.”
PASS TULIP components are differentiated from traditional top-loading implants by a thoughtful proprietary design that allows a single implant to perform multiple clinical functions through a simple one-step maneuver. The anticipated result is increased surgical efficiency in implant placement as well as a reduction in the inventory required for a case and associated processing costs.