Globe Newswire06.27.17
K2M Group Holdings Inc., a global developer of complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance, has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its MOJAVE PL 3D Expandable Interbody System. MOJAVE PL 3D is a first-to-market, 3D-printed expandable posterior-lumbar (PL) interbody system that features K2M’s Lamellar 3D Titanium Technology.
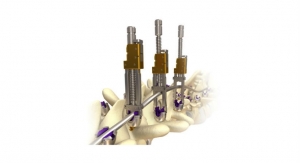
The MOJAVE PL 3D Expandable Interbody System is a fusion device designed to allow for independent control of the anterior and posterior height in the lumbar spine, a new capability not available with any other product in the market today, according to the company. Featuring infinite adjustment within the expansion range, the implant may be locked at any desired height and lordosis to aid in the restoration of sagittal balance.
“The ability to provide independent control of both the anterior and posterior height separately is desirable for the restoration of sagittal balance compared to existing devices that can’t independently adjust both anterior and posterior height,” said Steven Ludwig, M.D., an orthopedic spine surgeon and professor of orthopedics at the University of Maryland Medical Center in Baltimore.
K2M’s Lamellar 3D Titanium Technology uses an advanced 3D printing method to create structures that are impossible with traditional manufacturing techniques. Starting with a titanium powder, the MOJAVE PL 3D endplates are grown through the selective application of a high-energy laser beam, incorporating complex internal geometries and rough surface architecture that pre-clinical data have associated with bone growth activity. Lamellar 3D Titanium Technology incorporates a porous structure along with rough surfaces to allow the potential for bony integration throughout the endplates.
“We have developed internal 3D expertise that is allowing us to accelerate the rate of spinal innovation. As the first-ever, FDA-cleared, 3D-printed expandable interbody technology, MOJAVE PL 3D exemplifies our leadership in this space and provides surgeons the ability to expand the implant in-situ. This is our second family of products featuring Lamellar 3D Titanium Technology and builds upon the CASCADIA 3D family of static 3D-printed interbody cages,” said K2M President and CEO Eric Major. “Our innovation in 3D solutions and our focus on 3D spinal balance, demonstrated by the recent launch of our Balance ACS platform, solidifies our position as a market leader in the industry. We look forward to continuing to introduce new three-dimensional solutions to surgeons who treat patients with spinal disorders.”
Balance ACS (BACS) is a comprehensive platform applying three-dimensional solutions across the entire clinical care continuum to help drive quality outcomes in spine patients. BACS provides solutions focused on achieving balance of the spine by addressing each anatomical vertebral segment with a 360-degree approach to the axial, coronal, and sagittal planes, emphasizing Total Body Balance as an important component of surgical success.
K2M Group Holdings Inc. develops complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance. Since its inception, K2M has designed, developed, and commercialized complex spine and minimally invasive spine technologies and techniques used by spine surgeons to treat some of the most complicated spinal pathologies. K2M has leveraged these core competencies into Balance ACS, a platform of products, services, and research to help surgeons achieve three-dimensional spinal balance across the axial, coronal, and sagittal planes, with the goal of supporting the full continuum of care to facilitate quality patient outcomes. The company is based in Leesburg, Va.
The MOJAVE PL 3D Expandable Interbody System is a fusion device designed to allow for independent control of the anterior and posterior height in the lumbar spine, a new capability not available with any other product in the market today, according to the company. Featuring infinite adjustment within the expansion range, the implant may be locked at any desired height and lordosis to aid in the restoration of sagittal balance.
“The ability to provide independent control of both the anterior and posterior height separately is desirable for the restoration of sagittal balance compared to existing devices that can’t independently adjust both anterior and posterior height,” said Steven Ludwig, M.D., an orthopedic spine surgeon and professor of orthopedics at the University of Maryland Medical Center in Baltimore.
K2M’s Lamellar 3D Titanium Technology uses an advanced 3D printing method to create structures that are impossible with traditional manufacturing techniques. Starting with a titanium powder, the MOJAVE PL 3D endplates are grown through the selective application of a high-energy laser beam, incorporating complex internal geometries and rough surface architecture that pre-clinical data have associated with bone growth activity. Lamellar 3D Titanium Technology incorporates a porous structure along with rough surfaces to allow the potential for bony integration throughout the endplates.
“We have developed internal 3D expertise that is allowing us to accelerate the rate of spinal innovation. As the first-ever, FDA-cleared, 3D-printed expandable interbody technology, MOJAVE PL 3D exemplifies our leadership in this space and provides surgeons the ability to expand the implant in-situ. This is our second family of products featuring Lamellar 3D Titanium Technology and builds upon the CASCADIA 3D family of static 3D-printed interbody cages,” said K2M President and CEO Eric Major. “Our innovation in 3D solutions and our focus on 3D spinal balance, demonstrated by the recent launch of our Balance ACS platform, solidifies our position as a market leader in the industry. We look forward to continuing to introduce new three-dimensional solutions to surgeons who treat patients with spinal disorders.”
Balance ACS (BACS) is a comprehensive platform applying three-dimensional solutions across the entire clinical care continuum to help drive quality outcomes in spine patients. BACS provides solutions focused on achieving balance of the spine by addressing each anatomical vertebral segment with a 360-degree approach to the axial, coronal, and sagittal planes, emphasizing Total Body Balance as an important component of surgical success.
K2M Group Holdings Inc. develops complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance. Since its inception, K2M has designed, developed, and commercialized complex spine and minimally invasive spine technologies and techniques used by spine surgeons to treat some of the most complicated spinal pathologies. K2M has leveraged these core competencies into Balance ACS, a platform of products, services, and research to help surgeons achieve three-dimensional spinal balance across the axial, coronal, and sagittal planes, with the goal of supporting the full continuum of care to facilitate quality patient outcomes. The company is based in Leesburg, Va.