PR Newswire07.13.17
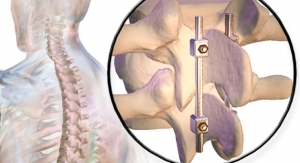
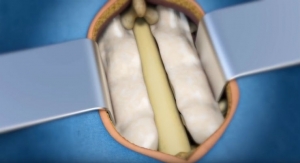
Pinnacle Spine Group LLC, the developer of in-situ graft delivery technology, has been granted a U.S. patent that meaningfully reinforces its exclusivity position with respect to its in-situ graft delivery technology. Patent No. 9,649,203, titled, "Methods of Post-Filling an Intervertebral Implant," like previously issued patents of the company, relates to the delivery of graft material to fill the internal chamber(s) of a spinal fusion device, while allowing for graft material to be in flush contact with the endplate surfaces of the adjacent vertebral bodies.
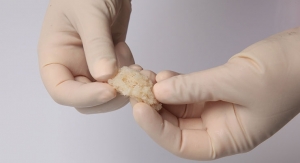
Pinnacle Spine Group developed and commercialized the concept of delivering bone graft material in-situ into an implanted device to fill the biologic void that remains with other spinal fusion systems. This latest patent follows the receipt of three earlier U.S. patents covering Pinnacle Spine’s InFill fusion technology. The company also owns several non-U.S. applications, including applications in Australia, Canada and Europe.
“Years ago, we identified a problem with traditional interbody fusion systems—that a biologic void remained between an implant and its two adjacent vertebrae,” said Zach Sowell, president of Pinnacle Spine Group. “We developed a solution and are thrilled to now have even stronger patent protection related to in-situ graft delivery directly to an implant, a technique whose value is being recognized and appreciated by the spinal fusion industry. This recognition is a testament to the technology’s usefulness and benefits, and we look forward to working with companies that are interested in adopting and using implant designs that require delivery of bone graft material in-situ.”
Pinnacle Spine’s InFill Fusion Systems include an array of interbody fusion devices engineered for easier insertion, reduced subsidence through maximum contact with the apophyseal ring, a generous bone grafting area, and compatibility with the InFill graft delivery system. The backbone of the technology is predicated on controlled and precise in-situ placement of bone graft material directly into the implanted device, to achieve maximum contact with the adjacent vertebral endplates.
Based in Dallas, Texas, Pinnacle Spine Group is a privately held company that develops and markets technologies to improve spinal fusion procedures. The company’s family of U.S. Food and Drug Administration-approved InFill Fusion systems provides for superior endplate-to-endplate contact of bone graft material and a complete fill of the disc space, achieved by post-filling the implants after they have been delivered into a targeted intervertebral disc space.
Pinnacle Spine Group developed and commercialized the concept of delivering bone graft material in-situ into an implanted device to fill the biologic void that remains with other spinal fusion systems. This latest patent follows the receipt of three earlier U.S. patents covering Pinnacle Spine’s InFill fusion technology. The company also owns several non-U.S. applications, including applications in Australia, Canada and Europe.
“Years ago, we identified a problem with traditional interbody fusion systems—that a biologic void remained between an implant and its two adjacent vertebrae,” said Zach Sowell, president of Pinnacle Spine Group. “We developed a solution and are thrilled to now have even stronger patent protection related to in-situ graft delivery directly to an implant, a technique whose value is being recognized and appreciated by the spinal fusion industry. This recognition is a testament to the technology’s usefulness and benefits, and we look forward to working with companies that are interested in adopting and using implant designs that require delivery of bone graft material in-situ.”
Pinnacle Spine’s InFill Fusion Systems include an array of interbody fusion devices engineered for easier insertion, reduced subsidence through maximum contact with the apophyseal ring, a generous bone grafting area, and compatibility with the InFill graft delivery system. The backbone of the technology is predicated on controlled and precise in-situ placement of bone graft material directly into the implanted device, to achieve maximum contact with the adjacent vertebral endplates.
Based in Dallas, Texas, Pinnacle Spine Group is a privately held company that develops and markets technologies to improve spinal fusion procedures. The company’s family of U.S. Food and Drug Administration-approved InFill Fusion systems provides for superior endplate-to-endplate contact of bone graft material and a complete fill of the disc space, achieved by post-filling the implants after they have been delivered into a targeted intervertebral disc space.