Integra LifeSciences Holdings Corporation10.11.17
Integra LifeSciences Holdings Corporation, a global medical technology company, announced the first Cadence Total Ankle System has been implanted by Dr. David Pedowitz with the Rothman Institute in Philadelphia, Pa., in the Cadence Total Ankle System non-randomized, prospective, multi-center post-market study.
"While this patient represents only one of many who have already benefited from the Cadence Total Ankle System, she is the first of my patients who will be a part of this important study, documenting the unique benefits of this new system in terms of ease of use, patient satisfaction, and survivability," said Pedowitz.
The primary objective of the study is to evaluate two-year implant survivorship in subjects who received the Cadence Total Ankle System for primary ankle arthroplasty. Implant survivorship will further be evaluated at five and 10 years post-operatively. All patients in the study will receive the Cadence Total Ankle System.
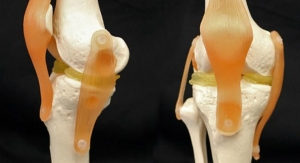
The Cadence Total Ankle System, is a new ankle prosthesis developed in partnership with four foot and ankle surgeons—Pedowitz; Dr. Tim Daniels (University of Toronto, Toronto, Canada); Dr. Selene Parekh (Duke University, Durham, N.C.) and Dr. Christopher Hyer (Orthopedic Foot & Ankle Center, Westerville, Ohio). To date, more than 350 Cadence total ankle procedures have been successfully performed in seven countries.
The Cadence system has garnered positive feedback among its users for advancements in implant and instrument design, along with a streamlined surgical technique. Cadence incorporates several features to accommodate various patient anatomies, reduce potential clinical complications, and address common challenges associated with ankle arthroplasty procedures. Key implant features include side-specific, anatomical tibial components, designed to avoid fibular impingement; and side-specific, anatomical talar components, designed to minimize resected talar bone and to preserve vascularity in the ankle. Bearing insert components are manufactured from a highly crosslinked ultra-high molecular weight polyethylene (HXL UHMWPE) for improved wear characteristics, and the system also features patent pending-biased sagittal profile options to address patients with subluxed talar anatomy.
Integra LifeSciences develops plastic and regenerative technologies in specialty surgical solutions, orthopedics and tissue-based science.
"While this patient represents only one of many who have already benefited from the Cadence Total Ankle System, she is the first of my patients who will be a part of this important study, documenting the unique benefits of this new system in terms of ease of use, patient satisfaction, and survivability," said Pedowitz.
The primary objective of the study is to evaluate two-year implant survivorship in subjects who received the Cadence Total Ankle System for primary ankle arthroplasty. Implant survivorship will further be evaluated at five and 10 years post-operatively. All patients in the study will receive the Cadence Total Ankle System.
The Cadence Total Ankle System, is a new ankle prosthesis developed in partnership with four foot and ankle surgeons—Pedowitz; Dr. Tim Daniels (University of Toronto, Toronto, Canada); Dr. Selene Parekh (Duke University, Durham, N.C.) and Dr. Christopher Hyer (Orthopedic Foot & Ankle Center, Westerville, Ohio). To date, more than 350 Cadence total ankle procedures have been successfully performed in seven countries.
The Cadence system has garnered positive feedback among its users for advancements in implant and instrument design, along with a streamlined surgical technique. Cadence incorporates several features to accommodate various patient anatomies, reduce potential clinical complications, and address common challenges associated with ankle arthroplasty procedures. Key implant features include side-specific, anatomical tibial components, designed to avoid fibular impingement; and side-specific, anatomical talar components, designed to minimize resected talar bone and to preserve vascularity in the ankle. Bearing insert components are manufactured from a highly crosslinked ultra-high molecular weight polyethylene (HXL UHMWPE) for improved wear characteristics, and the system also features patent pending-biased sagittal profile options to address patients with subluxed talar anatomy.
Integra LifeSciences develops plastic and regenerative technologies in specialty surgical solutions, orthopedics and tissue-based science.