PR Newswire11.01.17
Atlas Spine Inc., a high technology, spinal implant and instrumentation company based in Jupiter, Fla., has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its Ortus Expandable Posterior Lumbar Interbody System. The device is designed to be delivered using either minimally invasive or open surgical approach.
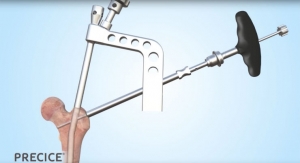
The Ortus PL is clearly differentiated from other expandable technology, according to Atlas Spine. For example, it has one of the smallest starting heights (6.5mm) of any expandable device on the market, and relies on a unique, patented design that allows for implant expansion while restoring natural anatomic alignment in a single step. The device also features an open architecture that allows the surgeon to pack ample graft material after the device is deployed and expanded. This is the first implant in a comprehensive line of expandable interbody intellectual property that utilizes anterior, PLIF, oblique, and minimally invasive approaches and devices that the company plans on launching.
“Atlas has a deep pipeline of expandable technology and the Ortus 510(k) clearance presents a tremendous opportunity to establish our company as an expandable interbody technology market leader. The Ortus platform provides options and capabilities for surgeons focused on restoring spinal balance that is yet to be provided by other devices and companies. Paired with a minimally invasive surgical approach, Ortus PL is a powerful tool for spine surgeons seeking reliable fusions with minimal complexity and post-operative morbidity. Furthermore, the Ortus PL platform is well suited for anterior, posterior, lateral and oblique approaches, in addition to MIS,” Atlas Spine Co-founder and Chief Technology Officer Matthew Baynham noted.
Atlas Spine CEO Douglass Watson added, “We are thrilled to be in the expandable posterior lumbar interbody market. The response from key, opinion-leading surgeons is tremendous. We are preparing to launch the Ortus PL interbody device with our distributor network in the near future and look forward to numerous additional differentiated device clearances over the next several quarters.”
The FDA notified the company on the first day of the North Atlantic Spine Society (NASS) meeting last week. The Ortus PL platform is Atlas Spine’s 12th product to receive 510(k) clearance by the FDA.
Atlas Spine develops spinal implants and instrument systems that treat a variety of pathologies. The company has over 50 issued and pending patents and currently distributes a number of interbody and posterior stabilization systems. It has also licensed technology to other companies in the spinal device market.
The Ortus PL is clearly differentiated from other expandable technology, according to Atlas Spine. For example, it has one of the smallest starting heights (6.5mm) of any expandable device on the market, and relies on a unique, patented design that allows for implant expansion while restoring natural anatomic alignment in a single step. The device also features an open architecture that allows the surgeon to pack ample graft material after the device is deployed and expanded. This is the first implant in a comprehensive line of expandable interbody intellectual property that utilizes anterior, PLIF, oblique, and minimally invasive approaches and devices that the company plans on launching.
“Atlas has a deep pipeline of expandable technology and the Ortus 510(k) clearance presents a tremendous opportunity to establish our company as an expandable interbody technology market leader. The Ortus platform provides options and capabilities for surgeons focused on restoring spinal balance that is yet to be provided by other devices and companies. Paired with a minimally invasive surgical approach, Ortus PL is a powerful tool for spine surgeons seeking reliable fusions with minimal complexity and post-operative morbidity. Furthermore, the Ortus PL platform is well suited for anterior, posterior, lateral and oblique approaches, in addition to MIS,” Atlas Spine Co-founder and Chief Technology Officer Matthew Baynham noted.
Atlas Spine CEO Douglass Watson added, “We are thrilled to be in the expandable posterior lumbar interbody market. The response from key, opinion-leading surgeons is tremendous. We are preparing to launch the Ortus PL interbody device with our distributor network in the near future and look forward to numerous additional differentiated device clearances over the next several quarters.”
The FDA notified the company on the first day of the North Atlantic Spine Society (NASS) meeting last week. The Ortus PL platform is Atlas Spine’s 12th product to receive 510(k) clearance by the FDA.
Atlas Spine develops spinal implants and instrument systems that treat a variety of pathologies. The company has over 50 issued and pending patents and currently distributes a number of interbody and posterior stabilization systems. It has also licensed technology to other companies in the spinal device market.