Business Wire02.07.18
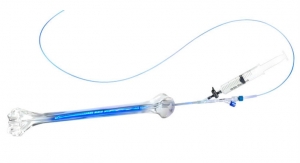
Orthofix International N.V., a global medical device company focused on musculoskeletal healing products, has announced the U.S. Food and Drug Administration 510(k) clearance and U.S. limited market launch of the FORZA XP Expandable Spacer System.
Designed to restore normal disc height in patients suffering from degenerative disc disease, the FORZA XP Expandable Spacer System can be expanded after insertion into the disc space in order to fit the patient’s anatomy. Made primarily of titanium alloy, these expandable interbodies are used for PLIF (Posterior Lumbar Interbody Fusion) and TLIF (Transforaminal Lumbar Interbody Fusion) procedures. Unlike the incremental expansion offered by some expandable interbody devices, the Orthofix System allows for a continuous controlled expansion and a custom fit to the disc space. Once the desired expansion is achieved the device features automatic locking which further adds to its ease of use.
“The FORZA XP Expandable Spacer System is a strong addition to our robust portfolio and makes Orthofix competitive in one of the fastest growing segments in spine,” said Ray Fujikawa, president of Orthofix Spine Fixation. “This new system offers a 6.5mm starting height and also enables surgeons to place bone graft material inside the device after implantation. The ability to pack our new expandable device with bone graft post expansion is extremely important to ensure proper contact of the material with the vertebral endplates.”
The FORZA XP Expandable Spacer System is indicated for spinal fusion procedures in skeletally mature patients with degenerative disc disease (DDD) at one or two contiguous levels in the lumbar spine. The FORZA XP Expandable Spacer System is intended for use with autograft and/or allograft comprised of cancellous and/or corticocancellous bone graft and supplemental fixation such as the Orthofix Trinity ELITE allograft tissue and the Firebird Spinal Fixation System.
Orthofix International N.V. is a global medical device company focused on musculoskeletal healing products and value-added services. The company’s mission is to improve patients’ lives by providing reconstruction and regenerative orthopedic and spine solutions to physicians worldwide. Headquartered in Lewisville, Texas, the company has four strategic business units: BioStim, Extremity Fixation, Spine Fixation, and Biologics.
Designed to restore normal disc height in patients suffering from degenerative disc disease, the FORZA XP Expandable Spacer System can be expanded after insertion into the disc space in order to fit the patient’s anatomy. Made primarily of titanium alloy, these expandable interbodies are used for PLIF (Posterior Lumbar Interbody Fusion) and TLIF (Transforaminal Lumbar Interbody Fusion) procedures. Unlike the incremental expansion offered by some expandable interbody devices, the Orthofix System allows for a continuous controlled expansion and a custom fit to the disc space. Once the desired expansion is achieved the device features automatic locking which further adds to its ease of use.
“The FORZA XP Expandable Spacer System is a strong addition to our robust portfolio and makes Orthofix competitive in one of the fastest growing segments in spine,” said Ray Fujikawa, president of Orthofix Spine Fixation. “This new system offers a 6.5mm starting height and also enables surgeons to place bone graft material inside the device after implantation. The ability to pack our new expandable device with bone graft post expansion is extremely important to ensure proper contact of the material with the vertebral endplates.”
The FORZA XP Expandable Spacer System is indicated for spinal fusion procedures in skeletally mature patients with degenerative disc disease (DDD) at one or two contiguous levels in the lumbar spine. The FORZA XP Expandable Spacer System is intended for use with autograft and/or allograft comprised of cancellous and/or corticocancellous bone graft and supplemental fixation such as the Orthofix Trinity ELITE allograft tissue and the Firebird Spinal Fixation System.
Orthofix International N.V. is a global medical device company focused on musculoskeletal healing products and value-added services. The company’s mission is to improve patients’ lives by providing reconstruction and regenerative orthopedic and spine solutions to physicians worldwide. Headquartered in Lewisville, Texas, the company has four strategic business units: BioStim, Extremity Fixation, Spine Fixation, and Biologics.