PR Newswire06.04.18
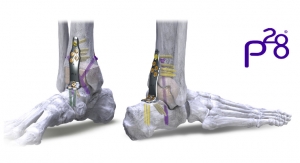
DT MedTech LLC announced that the first Hintermann Series H2 Total Ankle Replacement Systems have successfully been implanted in patients in the United States. Surgeries were performed, implanting the two-component, semi-constrained total ankle replacement prostheses, at The Johns Hopkins Hospital in Baltimore, Md., by Dr. James Ficke, director of Orthopaedic Surgery and Orthopaedist-in-Chief, and at Lakewood Ranch Medical Center in Sarasota, Fla., by Dr. James Cottom of Florida Orthopedic Foot and Ankle Center.
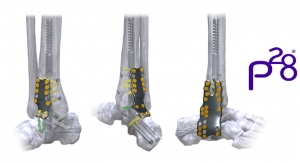
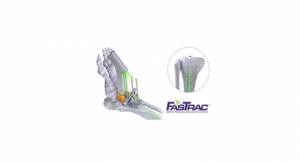
The Hintermann Series H2 Total Ankle Replacement System is indicated to replace painful arthritic ankle joints due to primary osteoarthritis, post-traumatic osteoarthritis, or arthritis secondary to inflammatory disease (e.g., rheumatoid arthritis, hemochromatosis). The H2 may also be used as a revision for failed ankle replacements or non-union and mal-union of ankle arthrodesis. It is intended for cement use only in the United States.
Prof. Beat Hintermann, world-renowned foot and ankle surgeon and developer of the H2, attended the surgeries and said, “I am extremely pleased to be here and observe the implantation of the first H2 ankles in the U.S. The surgeons did exemplary work and I am confident they will be very satisfied with their patients’ outcomes. The H2 design is based on the Hintermann H3* mobile-bearing ankle which has 18 years of positive clinical outcomes. I’m sure these ankles will prove to be the preferred ankle replacement prostheses for surgeons and patients in the United States and around the world.”
Cottom observed, “I believe this implant will be a game changer in ankle arthroplasty and think future clinical studies will demonstrate that it is in a class of its own.”
David Reicher, president and CEO of DTM, stated, “We could not be more pleased with the H2 surgeries that have recently taken place. With the successful results experienced from more than 40 European implants, we’re confident the U.S. success rates will be just as favorable. This exciting, complete ankle replacement system requires minimal bone resection and is designed to capture the axis of rotation which should result in highly reproducible outcomes. We feel it will significantly change the marketplace for primary and revision total ankle replacement surgeries on a global scale. Already available outside of the U.S., we expect a full U.S. product launch in September of this year, as we continue to focus on intense training for our surgeons.”
DT MedTech LLC is the parent company of DT MedTech International Limited and European Foot Platform S.A.R.L. DTM and its subsidiary companies maintain offices in Baltimore; Dublin, Ireland; Saint-Louis, France; and Liestal, Switzerland. As a member of the Data Trace family of businesses, leaders in scientific and medical publishing, surgical training, clinical trial management, medical malpractice insurance, and information services for more than 30 years, DTM provides surgical solutions for lower extremity surgeons with state-of-the-art devices such as The Hintermann Series Total Ankle Replacement Systems.
*The Hintermann Series H3 is not available for sale or distribution within the United States and its territories.
The Hintermann Series H2 Total Ankle Replacement System is indicated to replace painful arthritic ankle joints due to primary osteoarthritis, post-traumatic osteoarthritis, or arthritis secondary to inflammatory disease (e.g., rheumatoid arthritis, hemochromatosis). The H2 may also be used as a revision for failed ankle replacements or non-union and mal-union of ankle arthrodesis. It is intended for cement use only in the United States.
Prof. Beat Hintermann, world-renowned foot and ankle surgeon and developer of the H2, attended the surgeries and said, “I am extremely pleased to be here and observe the implantation of the first H2 ankles in the U.S. The surgeons did exemplary work and I am confident they will be very satisfied with their patients’ outcomes. The H2 design is based on the Hintermann H3* mobile-bearing ankle which has 18 years of positive clinical outcomes. I’m sure these ankles will prove to be the preferred ankle replacement prostheses for surgeons and patients in the United States and around the world.”
Cottom observed, “I believe this implant will be a game changer in ankle arthroplasty and think future clinical studies will demonstrate that it is in a class of its own.”
David Reicher, president and CEO of DTM, stated, “We could not be more pleased with the H2 surgeries that have recently taken place. With the successful results experienced from more than 40 European implants, we’re confident the U.S. success rates will be just as favorable. This exciting, complete ankle replacement system requires minimal bone resection and is designed to capture the axis of rotation which should result in highly reproducible outcomes. We feel it will significantly change the marketplace for primary and revision total ankle replacement surgeries on a global scale. Already available outside of the U.S., we expect a full U.S. product launch in September of this year, as we continue to focus on intense training for our surgeons.”
DT MedTech LLC is the parent company of DT MedTech International Limited and European Foot Platform S.A.R.L. DTM and its subsidiary companies maintain offices in Baltimore; Dublin, Ireland; Saint-Louis, France; and Liestal, Switzerland. As a member of the Data Trace family of businesses, leaders in scientific and medical publishing, surgical training, clinical trial management, medical malpractice insurance, and information services for more than 30 years, DTM provides surgical solutions for lower extremity surgeons with state-of-the-art devices such as The Hintermann Series Total Ankle Replacement Systems.
*The Hintermann Series H3 is not available for sale or distribution within the United States and its territories.