Ranica Arrowsmith, Associate Editor12.07.15
October saw a NASS preview of Nexxt Spine LLC’s NanoMatrixx, a new porous bioactive titanium material designed to actively participate in the intervertebral fusion process. The 3-D printed material is made to replicate the cellular structure of bone, and can be fashioned into any number of complex geometries with the desired level of elasticity.
Robert L. Wertz, vice president of new product development for the Noblesville, Ind.-based company, sat down with Orthopedic Design & Technology to discuss the product and what its future holds.
Arrowsmith: Could you describe the conception of NanoMatrixx?
Wertz: Prior to Nexxt Spine's inception in 2009, Founder and President Andy Elsbury provided contract manufacturing and design consultation to leading companies in the spine industry including Medtronic and DePuy. To this day he remains passionate about staying in front of cutting-edge manufacturing technologies and processes. NanoMatrixx is a porous bioactive titanium material manufactured to exacting specifications utilizing modern 3-D printing technology to replicate the cellular structure of cancellous bone. This process makes it possible to create any three dimensional complex structure or geometry that cannot be created by traditional orthopedic manufacturing processes. The inspiration for the NanoMatrixx design was fueled by increasing surgeon interest in better osteointegration of intervertebral spacers and reduction of material-related peri-inflammatory factors leading to fibrous tissue formation and encapsulation of devices.
Arrowsmith: What are its unique capabilities?
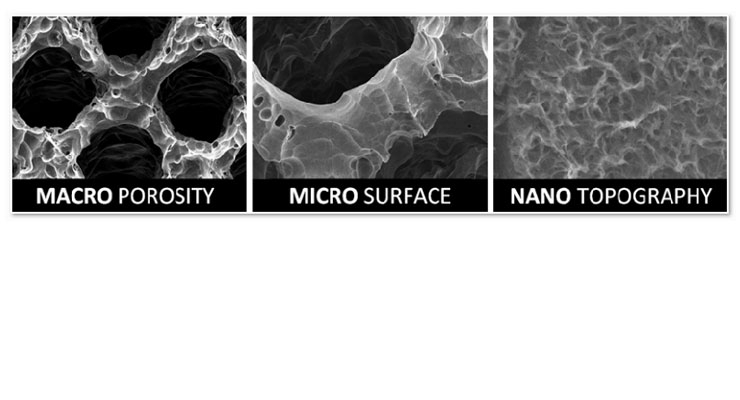
Wertz: A glimpse inside of the NanoMatrixx material reveals its 3-dimensional cellular architecture with a consistent 70 percent porosity. The proprietary scaffold provides an optimal biomechanical and biological environment for uninterrupted blood flow. Every surface of the NanoMatrixx material exhibits a micro-patterned nanotextured topography and is designed to elicit a superior osteogenic response. Titanium implants with roughened surfaces and titanium coated PEEK implant surfaces only achieve bone to implant contact or on-growth; however, NanoMatrixx’s consistent open and interconnected network of pores within well-established physiologically desirable ranges are designed to promote osteoconductivity and osteoinductivity, promoting bone on-growth and bone in-growth for total osseous integration. Bone has the potential to not only grow through the pores and around the struts, but also attach to the micro-patterned nanotextured strut surfaces throughout the device.
The cancellous-like interconnected macro porosity, electrically charged micro-roughened surface, bioactive nanotopography, and modulus of elasticity, provide a unique combination of attributes that contribute to the material’s proposed osteoconductive and osteoinductive properties allowing it to participate in the fusion process.
Arrowsmith: What are the plans for the commercialization and future development of the product?
Wertz: We are currently working with recognized key opinion leaders and experts in the area of spine surgery, tissue engineering and surface technology to develop a robust research agenda to exploit innovation utilizing NanoMatrixx as the foundation for clinical applications with potential game changing implications. In Q1/2016 we will be submitting our first group of intervertebral devices to the FDA for 510(k) clearance. The remainder of the products completing a comprehensive intervertebral product line will move through the FDA at a regular pace following our initial product releases.
Arrowsmith: Is the product marketed domestically only, or also internationally?
Wertz: Our current distribution network throughout the United States is anxiously awaiting the NanoMatrixx Interbody product launch. We are also focused on establishing international distribution by the end of 2017. Several key territories have been identified and well established distribution partners are currently on our radar.
Arrowsmith: How did the preview at NASS go? Where else will Nexxt Spine be previewing the product?
Wertz: This was my 10th NASS appearance serving in a product development capacity and Nexxt Spine’s first as an exhibitor. It was surprisingly the busiest show I’ve covered due to the overwhelming amount of interest surrounding NanoMatrixx from surgeons and medical device distributors. NASS has been somewhat disappointing over the past few years from an innovation standpoint according to many surgeons we’ve spoken to and our forthcoming technology piqued an enormous amount of interest with a never ending flow of traffic to our booth at the show. We are in the process of finalizing our 2016 conference calendar.
In the meantime, anyone looking to learn more about NanoMatrixx or Nexxt Spine can reach us directly at (317) 436-7801 or info@nexxtspine.com.
Robert L. Wertz, vice president of new product development for the Noblesville, Ind.-based company, sat down with Orthopedic Design & Technology to discuss the product and what its future holds.
Arrowsmith: Could you describe the conception of NanoMatrixx?
Wertz: Prior to Nexxt Spine's inception in 2009, Founder and President Andy Elsbury provided contract manufacturing and design consultation to leading companies in the spine industry including Medtronic and DePuy. To this day he remains passionate about staying in front of cutting-edge manufacturing technologies and processes. NanoMatrixx is a porous bioactive titanium material manufactured to exacting specifications utilizing modern 3-D printing technology to replicate the cellular structure of cancellous bone. This process makes it possible to create any three dimensional complex structure or geometry that cannot be created by traditional orthopedic manufacturing processes. The inspiration for the NanoMatrixx design was fueled by increasing surgeon interest in better osteointegration of intervertebral spacers and reduction of material-related peri-inflammatory factors leading to fibrous tissue formation and encapsulation of devices.
Arrowsmith: What are its unique capabilities?
Wertz: A glimpse inside of the NanoMatrixx material reveals its 3-dimensional cellular architecture with a consistent 70 percent porosity. The proprietary scaffold provides an optimal biomechanical and biological environment for uninterrupted blood flow. Every surface of the NanoMatrixx material exhibits a micro-patterned nanotextured topography and is designed to elicit a superior osteogenic response. Titanium implants with roughened surfaces and titanium coated PEEK implant surfaces only achieve bone to implant contact or on-growth; however, NanoMatrixx’s consistent open and interconnected network of pores within well-established physiologically desirable ranges are designed to promote osteoconductivity and osteoinductivity, promoting bone on-growth and bone in-growth for total osseous integration. Bone has the potential to not only grow through the pores and around the struts, but also attach to the micro-patterned nanotextured strut surfaces throughout the device.
The cancellous-like interconnected macro porosity, electrically charged micro-roughened surface, bioactive nanotopography, and modulus of elasticity, provide a unique combination of attributes that contribute to the material’s proposed osteoconductive and osteoinductive properties allowing it to participate in the fusion process.
Arrowsmith: What are the plans for the commercialization and future development of the product?
Wertz: We are currently working with recognized key opinion leaders and experts in the area of spine surgery, tissue engineering and surface technology to develop a robust research agenda to exploit innovation utilizing NanoMatrixx as the foundation for clinical applications with potential game changing implications. In Q1/2016 we will be submitting our first group of intervertebral devices to the FDA for 510(k) clearance. The remainder of the products completing a comprehensive intervertebral product line will move through the FDA at a regular pace following our initial product releases.
Arrowsmith: Is the product marketed domestically only, or also internationally?
Wertz: Our current distribution network throughout the United States is anxiously awaiting the NanoMatrixx Interbody product launch. We are also focused on establishing international distribution by the end of 2017. Several key territories have been identified and well established distribution partners are currently on our radar.
Arrowsmith: How did the preview at NASS go? Where else will Nexxt Spine be previewing the product?
Wertz: This was my 10th NASS appearance serving in a product development capacity and Nexxt Spine’s first as an exhibitor. It was surprisingly the busiest show I’ve covered due to the overwhelming amount of interest surrounding NanoMatrixx from surgeons and medical device distributors. NASS has been somewhat disappointing over the past few years from an innovation standpoint according to many surgeons we’ve spoken to and our forthcoming technology piqued an enormous amount of interest with a never ending flow of traffic to our booth at the show. We are in the process of finalizing our 2016 conference calendar.
In the meantime, anyone looking to learn more about NanoMatrixx or Nexxt Spine can reach us directly at (317) 436-7801 or info@nexxtspine.com.