Sean Fenske, Editor03.14.16
It’s not often that you hear of a true treasure being “rescued” from a bankruptcy auction (especially in the medtech space), but in the case of TranS1, that’s exactly what its CEO, Jeff Schell, made sure happened. Recognizing the importance of the technology for several years prior, Schell moved quickly when he learned of Baxano Surgical’s impending exit from the industry. As a result, he secured a technology for his company that he viewed as a true disruptive innovation within the spine fusion sector.
“AxiaLIF provides a strong foundation at the L5-S1 joint to enable many patients to return to active lifestyles. AxiaLIF remains a key advance in spine surgery over the past twenty years. The company continues to develop a robust product portfolio around AxiaLIF as its core product,” explained Schell during an interview with ODT.
In the following presentation, Schell explains the strategy he’s taken with regard to securing the AxiaLIF technology, the opportunities it offers, how his company is continuing to support it, and what’s coming in the years ahead.
Sean Fenske: Please tell me a bit about TranS1.
Jeff Schell:

Jeff Schell is the CEO of TranS1.
TranS1 is a spinal device company dedicated to improving patient outcomes. Headquartered in Denver, Colo., the company features a product line that provides a foundation for living well. Driven by surgeon innovation, the team includes experienced R&D, sales, legal, and management professionals who focus on resilient and continuous training, employing best-in-class training methods that ultimately benefit patients. In particular, the R&D focus of the company has led to its hiring a team of engineers, Ph.D.s, and other experts to assist with development of paradigm-shifting technologies in spine surgery.
Fenske: The company “salvaged” the AxiaLIF technology from a bankruptcy auction. Can you tell me about that experience?
Schell: I learned about the impending dissolution of Baxano Surgical in 2014. For several years prior, I followed the AxiaLIF product very closely, knowing that its unique approach represented one of the most significant advances in spine surgery over the past few decades. Many surgeons around the country continue to share this sentiment. As a patent lawyer, I also knew of the true value of the intellectual property portfolio associated with AxiaLIF. More than 100 patents of significant value and importance suddenly became available during the dissolution of Baxano. The product had real importance for surgeons and patients alike.
I started several companies in Denver during 2013-2014, including Rocky Mountain Patent and Quandary Medical, which began commercializing its own medical devices. When Baxano announced its dissolution, I saw the need to act quickly to acquire the AxiaLIF technology. I, along with many in the spine surgery community, viewed the technology as, by far, the most important piece of Baxano’s portfolio. Over December 2014 and January 2015, I worked to secure a financing facility of several million dollars to both acquire and support the AxiaLIF procedure, along with our other R&D initiatives. I have heard from several surgeons, investors, and spine company executives that they had significant interest in acquiring the product portfolio; I feel fortunate that TranS1’s vice president, Walker Wells, was able to win the auction for the AxiaLIF while at the same time negotiating an excellent price point. We were fortunate to acquire the AxiaLIF technology and its IP at a price that enabled us to have ample capital to re-energize the supply chain, compliance, enforcement, marketing, public relations, and, most importantly, surgeon support operations for AxiaLIF.
Fenske: Was this just a fortunate situation or is there strong interest in securing technology via this method?
Schell: I view AxiaLIF as one of the most important options for treatment of degenerative disc disease and spondylolythesis at the L5-S1 joint. I have been involved in healthcare and surgical operations for over a decade. During that time, I developed relationships with many spine surgeons. Most of those surgeons share their opinion that AxiaLIF represents one of the most important options for treatment of pathology at L5-S1. The technology has fallen into good hands.
To answer your question more specifically though, we are not in a habit of acquiring IP and technology from bankruptcy proceedings, but we would have gone anywhere in the world to buy the AxiaLIF. It was too promising of an approach—too disruptive to the status quo of L5-S1 fusion—for us to miss the opportunity.
Fenske: Tell me more about the AxiaLIF technology. How is it different from other solutions indicated for the treatment of the lumbar region?
Schell: As numerous peer-reviewed studies demonstrate, AxiaLIF accomplishes a 94 percent fusion rate while minimizing blood loss, lowering time needed in the operating room, and reducing the length of stay in the hospital compared to the alternatives at L5-S1. Properly selected patients can regularly receive AxiaLIF and leave the hospital or surgery center the same day, which makes sense as it requires the smallest incision.
Unlike other procedures, some of which usually require the removal of an entire facet joint (TLIF); retraction of the dura, which can lead to nerve injury (TLIF/PLIF); or dissection through the abdomen, which can lead to vascular perforation (ALIF); the AxiaLIF’s alternative pre-sacral approach preserves bony supportive structures and avoids other complications. In fact, a retrospective review of over 9,000 cases indicated that AxiaLIF’s complication rate is less than two percent—much less than alternatives at L5-S1. The same study found that the main unique risk associated with AxiaLIF—bowel perforation—had a risk of less than one percent. The risk profile compared to the efficacy of the other L5-S1 spinal fusion procedures is well-studied (with over 75 peer-reviewed articles) and very favorable.
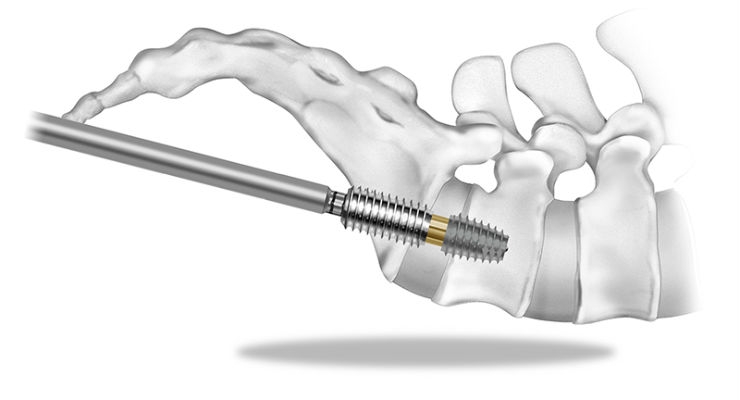
Briefly, the surgical steps of the procedure differ from other procedures. To access the L5-S1 area for AxiaLIF fusion, a surgeon makes a small incision next to the tailbone. The surgeon then follows the anterior surface of the tailbone with a dissector. Cannulas are placed to provide a working channel for the surgeon. The surgeon then utilizes the working channel to drill through the sacrum into the L5-S1 space. Our discectomy tools then allow for removal of disc material. The surgeon can then place bone graft and the AxiaLIF implants across the L5-S1 space to allow for fusion. The primary difference of the procedure is the approach. Many of the steps associated with the procedure, such as access, discectomy, placement of bone graft, and placement of an implant, are similar or identical to the other procedures, though those procedures take a different approach angle to the spine.
[Editor's Note: The following video offers an expanded view of surgical procedure associated with the AxiaLIF. Interview continues after it.]
Fenske: Could the technology be adapted for other orthopedic indications/procedures?
Schell: Yes. Absolutely. TranS1 has a very strong research and development team, featuring several in-house engineers, Ph.D. researchers, and patent attorneys. We have filed many patent applications related to other adaptations of the pre-sacral approach and other approaches in the last year. We believe that the pre-sacral approach represents one of the most important opportunities for advancement in orthopedic surgery and for other surgical fields.
Fenske: With the development of new materials, a more natural approach could be indicated for this region. Can the technology be adapted to serve that type of procedure?
Schell: Definitely. TranS1 has taken a leading role in the exploration of other material alternatives, including 3D-printed titanium, impregnated PEEK, and carbon-based implants. We have filed patent applications related to these and other technologies. We do believe that AxiaLIF and our other proprietary technologies are adaptable to a wide range of procedures.
Fenske: What’s the next step for the company and this technology?
Schell: TranS1 will continue upon its mission to minimize tissue trauma associated with approaches to spinal fusion. We are working on multiple fronts, including continuing to explore multiple acquisition opportunities. Our R&D team continues to work with spine surgeons around the country to advance new ideas for surgery. We have recognized and experienced that in the past, companies in the industry often marginalize surgeons when approaching new technologies; we hope to reverse this trend. Our combined surgeon/R&D efforts have yielded over thirty filed patent applications in the past eighteen months. As a patent attorney, I believe that owning the rights to new approaches and technologies remains fundamentally important to the growth of our company.
I expect that we will have filed over 50 patent applications targeting important relevant technologies by the end of 2016. While our primary objective always has been to secure and support the important AxiaLIF technology and to continue development of paradigm-shifting new technologies, as a patent attorney with a skilled team of patent litigators in my employ, patent enforcement where necessary remains a key aspect of our strategy.
Most importantly, however, we will engage surgeons to work with us to contribute their ideas to develop new technologies. Quandary Medical was conceived as a company to advance spine surgeon ideas. Increasingly over the next year, while continuing to strongly supporting AxiaLIF, we will shift our focus to advancing new, paradigm shifting surgical technologies and techniques.
Fenske: What’s on the horizon for spinal fusion in five to ten years?
Schell: Minimizing tissue trauma associated with access to the spine. Given that the pre-sacral approach is often considered the most minimally invasive access path to the spine, leading to many well-documented advantages in peer-reviewed literature, our company is well-positioned to leverage this trend at L5-S1.
Related to the reduction in tissue trauma associated with approaching the spine in new technologies, spine surgery will increasingly take place on an outpatient basis. Outpatient procedures reduce costs and recovery time for patients. With the right patient selection, spinal fusion can be performed on an outpatient basis safely and effectively.
Unfortunately, a variety of special interests and economic conflicts will remain at play, particularly in orthopedics. Payors, large manufacturers, hospitals, and other players continue to fight over their part of a shrinking pool of funds available for healthcare. Cost reduction will continue to play a role, and, fortunately for TranS1, a recent Journal of Managed Care article demonstrated cost savings of several thousands of dollars per episode of care related to L5-S1 fusion.
Related to cost reductions, I expect to continue to see conflicts of interest and anticompetitive behavior from firms with the most to lose playing a greater role in choice-of-care decisions. These cost and market share considerations will crowd out more important considerations for choice of care, including, “Will this procedure lead to a better outcome for a particular patient?” Companies will increasingly need to watch out for anticompetitive behavior and allocate resources to combat interests illegally causing market exclusion.
Fenske: Before closing, do you have any additional thoughts or comments you’d like to share?
Schell: Our company’s motto, “Building a Strong Foundation,” applies not only to the products we produce but also to the company itself. For several years prior to our acquisition of AxiaLIF and the TranS1 brand, TranS1 had been thought of as a “passion story” in spine. It was a startup company with an incredible technology that resonated well with surgeons and offered a meaningful alternative at L5-S1. Over the last year, we have re-ignited that vision. We have created an incredibly talented team to build upon an established technology. Our team shares the vision that access to the spine to accomplish effective fusion need not require a great amount of trauma to the surrounding tissues. I am excited to coordinate our vision and talented team with surgeons to advance new, paradigm-shifting approaches to treatments of the spine.
“AxiaLIF provides a strong foundation at the L5-S1 joint to enable many patients to return to active lifestyles. AxiaLIF remains a key advance in spine surgery over the past twenty years. The company continues to develop a robust product portfolio around AxiaLIF as its core product,” explained Schell during an interview with ODT.
In the following presentation, Schell explains the strategy he’s taken with regard to securing the AxiaLIF technology, the opportunities it offers, how his company is continuing to support it, and what’s coming in the years ahead.
Sean Fenske: Please tell me a bit about TranS1.
Jeff Schell:
Jeff Schell is the CEO of TranS1.
Fenske: The company “salvaged” the AxiaLIF technology from a bankruptcy auction. Can you tell me about that experience?
Schell: I learned about the impending dissolution of Baxano Surgical in 2014. For several years prior, I followed the AxiaLIF product very closely, knowing that its unique approach represented one of the most significant advances in spine surgery over the past few decades. Many surgeons around the country continue to share this sentiment. As a patent lawyer, I also knew of the true value of the intellectual property portfolio associated with AxiaLIF. More than 100 patents of significant value and importance suddenly became available during the dissolution of Baxano. The product had real importance for surgeons and patients alike.
I started several companies in Denver during 2013-2014, including Rocky Mountain Patent and Quandary Medical, which began commercializing its own medical devices. When Baxano announced its dissolution, I saw the need to act quickly to acquire the AxiaLIF technology. I, along with many in the spine surgery community, viewed the technology as, by far, the most important piece of Baxano’s portfolio. Over December 2014 and January 2015, I worked to secure a financing facility of several million dollars to both acquire and support the AxiaLIF procedure, along with our other R&D initiatives. I have heard from several surgeons, investors, and spine company executives that they had significant interest in acquiring the product portfolio; I feel fortunate that TranS1’s vice president, Walker Wells, was able to win the auction for the AxiaLIF while at the same time negotiating an excellent price point. We were fortunate to acquire the AxiaLIF technology and its IP at a price that enabled us to have ample capital to re-energize the supply chain, compliance, enforcement, marketing, public relations, and, most importantly, surgeon support operations for AxiaLIF.
Fenske: Was this just a fortunate situation or is there strong interest in securing technology via this method?
Schell: I view AxiaLIF as one of the most important options for treatment of degenerative disc disease and spondylolythesis at the L5-S1 joint. I have been involved in healthcare and surgical operations for over a decade. During that time, I developed relationships with many spine surgeons. Most of those surgeons share their opinion that AxiaLIF represents one of the most important options for treatment of pathology at L5-S1. The technology has fallen into good hands.
To answer your question more specifically though, we are not in a habit of acquiring IP and technology from bankruptcy proceedings, but we would have gone anywhere in the world to buy the AxiaLIF. It was too promising of an approach—too disruptive to the status quo of L5-S1 fusion—for us to miss the opportunity.
Fenske: Tell me more about the AxiaLIF technology. How is it different from other solutions indicated for the treatment of the lumbar region?
Schell: As numerous peer-reviewed studies demonstrate, AxiaLIF accomplishes a 94 percent fusion rate while minimizing blood loss, lowering time needed in the operating room, and reducing the length of stay in the hospital compared to the alternatives at L5-S1. Properly selected patients can regularly receive AxiaLIF and leave the hospital or surgery center the same day, which makes sense as it requires the smallest incision.
Unlike other procedures, some of which usually require the removal of an entire facet joint (TLIF); retraction of the dura, which can lead to nerve injury (TLIF/PLIF); or dissection through the abdomen, which can lead to vascular perforation (ALIF); the AxiaLIF’s alternative pre-sacral approach preserves bony supportive structures and avoids other complications. In fact, a retrospective review of over 9,000 cases indicated that AxiaLIF’s complication rate is less than two percent—much less than alternatives at L5-S1. The same study found that the main unique risk associated with AxiaLIF—bowel perforation—had a risk of less than one percent. The risk profile compared to the efficacy of the other L5-S1 spinal fusion procedures is well-studied (with over 75 peer-reviewed articles) and very favorable.
Briefly, the surgical steps of the procedure differ from other procedures. To access the L5-S1 area for AxiaLIF fusion, a surgeon makes a small incision next to the tailbone. The surgeon then follows the anterior surface of the tailbone with a dissector. Cannulas are placed to provide a working channel for the surgeon. The surgeon then utilizes the working channel to drill through the sacrum into the L5-S1 space. Our discectomy tools then allow for removal of disc material. The surgeon can then place bone graft and the AxiaLIF implants across the L5-S1 space to allow for fusion. The primary difference of the procedure is the approach. Many of the steps associated with the procedure, such as access, discectomy, placement of bone graft, and placement of an implant, are similar or identical to the other procedures, though those procedures take a different approach angle to the spine.
[Editor's Note: The following video offers an expanded view of surgical procedure associated with the AxiaLIF. Interview continues after it.]
Fenske: Could the technology be adapted for other orthopedic indications/procedures?
Schell: Yes. Absolutely. TranS1 has a very strong research and development team, featuring several in-house engineers, Ph.D. researchers, and patent attorneys. We have filed many patent applications related to other adaptations of the pre-sacral approach and other approaches in the last year. We believe that the pre-sacral approach represents one of the most important opportunities for advancement in orthopedic surgery and for other surgical fields.
Fenske: With the development of new materials, a more natural approach could be indicated for this region. Can the technology be adapted to serve that type of procedure?
Schell: Definitely. TranS1 has taken a leading role in the exploration of other material alternatives, including 3D-printed titanium, impregnated PEEK, and carbon-based implants. We have filed patent applications related to these and other technologies. We do believe that AxiaLIF and our other proprietary technologies are adaptable to a wide range of procedures.
Fenske: What’s the next step for the company and this technology?
Schell: TranS1 will continue upon its mission to minimize tissue trauma associated with approaches to spinal fusion. We are working on multiple fronts, including continuing to explore multiple acquisition opportunities. Our R&D team continues to work with spine surgeons around the country to advance new ideas for surgery. We have recognized and experienced that in the past, companies in the industry often marginalize surgeons when approaching new technologies; we hope to reverse this trend. Our combined surgeon/R&D efforts have yielded over thirty filed patent applications in the past eighteen months. As a patent attorney, I believe that owning the rights to new approaches and technologies remains fundamentally important to the growth of our company.
I expect that we will have filed over 50 patent applications targeting important relevant technologies by the end of 2016. While our primary objective always has been to secure and support the important AxiaLIF technology and to continue development of paradigm-shifting new technologies, as a patent attorney with a skilled team of patent litigators in my employ, patent enforcement where necessary remains a key aspect of our strategy.
Most importantly, however, we will engage surgeons to work with us to contribute their ideas to develop new technologies. Quandary Medical was conceived as a company to advance spine surgeon ideas. Increasingly over the next year, while continuing to strongly supporting AxiaLIF, we will shift our focus to advancing new, paradigm shifting surgical technologies and techniques.
Fenske: What’s on the horizon for spinal fusion in five to ten years?
Schell: Minimizing tissue trauma associated with access to the spine. Given that the pre-sacral approach is often considered the most minimally invasive access path to the spine, leading to many well-documented advantages in peer-reviewed literature, our company is well-positioned to leverage this trend at L5-S1.
Related to the reduction in tissue trauma associated with approaching the spine in new technologies, spine surgery will increasingly take place on an outpatient basis. Outpatient procedures reduce costs and recovery time for patients. With the right patient selection, spinal fusion can be performed on an outpatient basis safely and effectively.
Unfortunately, a variety of special interests and economic conflicts will remain at play, particularly in orthopedics. Payors, large manufacturers, hospitals, and other players continue to fight over their part of a shrinking pool of funds available for healthcare. Cost reduction will continue to play a role, and, fortunately for TranS1, a recent Journal of Managed Care article demonstrated cost savings of several thousands of dollars per episode of care related to L5-S1 fusion.
Related to cost reductions, I expect to continue to see conflicts of interest and anticompetitive behavior from firms with the most to lose playing a greater role in choice-of-care decisions. These cost and market share considerations will crowd out more important considerations for choice of care, including, “Will this procedure lead to a better outcome for a particular patient?” Companies will increasingly need to watch out for anticompetitive behavior and allocate resources to combat interests illegally causing market exclusion.
Fenske: Before closing, do you have any additional thoughts or comments you’d like to share?
Schell: Our company’s motto, “Building a Strong Foundation,” applies not only to the products we produce but also to the company itself. For several years prior to our acquisition of AxiaLIF and the TranS1 brand, TranS1 had been thought of as a “passion story” in spine. It was a startup company with an incredible technology that resonated well with surgeons and offered a meaningful alternative at L5-S1. Over the last year, we have re-ignited that vision. We have created an incredibly talented team to build upon an established technology. Our team shares the vision that access to the spine to accomplish effective fusion need not require a great amount of trauma to the surrounding tissues. I am excited to coordinate our vision and talented team with surgeons to advance new, paradigm-shifting approaches to treatments of the spine.