08.09.16
$9.9 Billion
KEY EXECUTIVES:
Kevin A. Lobo, Chairman and CEO
Lonny J. Carpenter, Group President, Global Quality and Business Operations
Glenn S. Boehnlein, VP, Chief Financial Officer
David K. Floyd, Group President, Orthopaedics
Timothy J. Scannell, Group President, MedSurg & Neurotechnology
J. Andrew Pierce, President, Endoscopy
Spencer S. Stiles, President, Instruments
William J. Huffnagle, President, Reconstructive
Xavier Berling, President, Trauma and Extremities
Mark H. Paul, President, Neurovascular
Bradford L. Saar, President, Medical
Bradley W. Paddock, President, Spine
Wayne D. Dahlberg, President, Performance Solutions
NUMBER OF EMPLOYEES: 27,000
HEADQUARTERS: Kalamazoo, Mich.
Beth Roche always considered herself a “mover.”
“I’m a person who likes to get up and get it done,” she told a Chicago Tribune reporter several years ago.
Imagine her level of frustration, then, as she struggled to recover from injuries she sustained in the 2013 Boston Marathon bombing. The Highland, Ind., mother and avid runner was among hundreds wounded in the terrorist attack, suffering a compound fracture of her left tibia and a shattered kneecap.
Roche was in Beantown on the day of the blast to watch her daughter Rebecca run the marathon. The first of two homemade bombs exploded shortly after Rebecca crossed the finish line, spraying a leg-level pattern of shrapnel that killed three spectators and peeled back the skin on Roche’s knee “like a sardine can,” leaving the bones exposed.
Oddly, Roche felt no pain. Panic ensued as bystanders stampeded toward a nearby sports store; the wail of emergency sirens filled the air, mixing with piercing screams from the injured and frightened. “Run!” someone yelled to Roche.
“I can’t run,” she thought, unable even to stand.
Within minutes, public safety and rescue personnel covered Roche with a blanket and treated her bleeding wound with a tourniquet. Paramedics rushed her to Tufts Medical Center, where orthopedic trauma surgeons cleaned her wounds of debris (fishing out bits of the black canvas backpack that housed one of the bombs) and reconstructed her left knee in a four-hour surgery.
Although Roche lost a significant amount of skin, muscle, bone, and tendon in the attack, blood flowed to her lower leg and foot, enabling surgeons to save the limb. Dr. Scott Ryan, orthopedic trauma chief at Tufts, stabilized Roche’s damaged bones with rods and screws, and held her newly repaired (but still fragile) knee firmly in place with an external frame from Stryker Corp.
Composed of lightweight carbon-based materials, the Hoffmann II MRI External Fixation System is designed to provide patient comfort and facilitate fracture management and stabilization.
Cleared for magnetic resonance imaging use under prescribed conditions, the Hoffmann frame employs “snap-fit” technology, allowing doctors to conduct postoperative corrections and treatments with the device in place. Clinicians also can build the frame to treat fractures close to a joint (as in Roche’s case) or stay clear of damaged soft-tissue areas.
After undergoing three surgeries to remove scar tissue and replace lost ligaments, as well as months of grueling physical therapy, Roche has finally regained flexibility in her knee. She’s exceeded her doctors’ expectations for a modest recovery, reclaiming the ability to bend her knee 145 degrees (Ryan had predicted 90-degree flexibility). But chronic pain forces Roche to rethink stairs and occasionally doubt her ability to resume an active lifestyle.
Nevertheless, she refuses to give up hope.
“My life is a race now,” Roche said last fall. “It’s a physical, mental race to get back to normal. What my normal was before the bomb.”
Nora Kasapligil, 21, has already won that race. Her life returned to normal remarkably quickly—within a day of suffering an ischemic stroke in her Sonoma State University dorm room. Kasapligil awoke on June 5, 2015, with a severe headache, and after getting out of bed, walked straight into a wall. Luckily, her roommate noticed Kasapligil’s odd speech and movements and quickly summoned help, suspecting a stroke.
“I think I tried saying, ‘I have to go to work,’ and instead of it coming out like that, I said, ‘I have to go, go, go, go to work,’ ” the young woman recently recounted to a Northern California television station.
Medical scans at a local hospital eventually confirmed the roommate’s suspicions: a clot was blocking the blood supply to the right side of Kasapligil’s brain. But since it formed in her sleep, doctors weren’t sure they could use tissue plasminogen activator, or tPA, the U.S. Food and Drug Administration (FDA)-approved clot-busting drug that must be given intravenously with 4.5 hours to be effective. “After four and a half hours, the data tell us the risk of bleeding in the brain is greater than providing any benefit of opening the [blood] vessel,” explained Joey D. English, M.D., director of Neurointerventional Services at California Pacific Medical Center in San Francisco.
After confirming the affected brain tissue was still surviving, English and his colleagues treated Kasapligil’s stroke by removing her blood clot with a device developed by Stryker. The company’s Trevo XP ProVue Retriever clears blocked vessels using an expandable wire mesh stent that snares the clot and drags it out of the body (via catheters) through the femoral artery. The FDA-cleared device is billed as the world’s first stent retriever with full-length visibility, giving surgeons greater control and real-time information during placement, integration, and clot removal.
Within an hour of treatment with the Trevo XP device, Kasapligil’s left-side paralysis began to dissipate; her facial weakness and slurred speech disappeared a day later.
“The new stent retrieval devices for treating large-vessel occlusions are pretty remarkable advances,” English said. “I hate to think what Nora’s outcome would have been without this new technology. She’s living proof that Trevo has changed the way we treat stroke.”
Indeed, stent retrievers like the Trevo are rewriting the rules of ischemic stroke treatment. Last summer, the American Heart Association (AHA) and American Stroke Association issued guidelines recommending the use of stent retrievers in concert with tPA after five studies published in the New England Journal of Medicine found the devices reduced disability, improved neurological function, shortened recovery time, and increased the rate at which stroke survivors regain function. The guidelines advised the stent retrieval procedure be performed on adult patients within six hours of acute stroke symptoms, and only after imaging proves the affected cranial area is not permanently damaged. The endorsement was the AHA’s first in two decades.
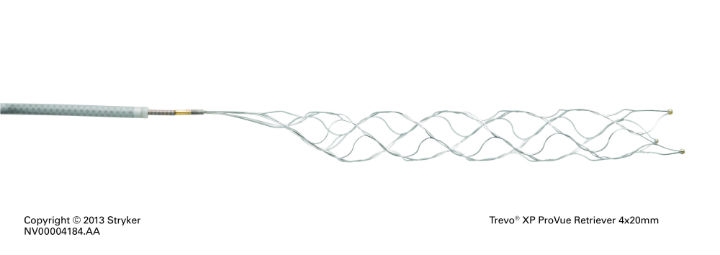
The Trevo XP ProVue Retriever was one of the key products used in the clinical trials that prompted the American Heart Association and American Stroke Association to issue new guidelines last year recommending endovascular therapy with stent retrievers for ischemic stroke patients. (Courtesy of Stryker Corp.)
Though stent retrievers are available at more than 1,000 U.S. stroke centers and hospitals, only about 13,000 procedures are performed annually—mostly at comprehensive stroke centers—because they require specialized training. The devices are now so technically sophisticated that properly trained interventional neuroradiologists could regularly remove clots within an hour, with the best treatment time of just under 20 minutes from the initial computed tomography scan.
Despite their promise, however, retrievers like the ProVue will most likely remain on the periphery of standardized treatment without significant market evolution. “...an impressive amount of strong clinical data has been released supporting the use of device-based treatment of acute ischemic stroke,” Katherine Owen, Stryker’s vice president of strategy and investor relations, noted to analysts during a first-quarter earnings conference call last spring. “We have pioneered this space in our unique and differentiated products. We expect the market will take time to evolve, which will require the automation of EMS transport and inter-hospital transfers, establishment of clinical guidelines, position incentives, and investment in new human and physical capital to absorb new patient volume. Clearly as the data comes out, it reinforces our excitement from the ischemic stroke market and its longer-term revenue potential.”
The market’s short-term potential isn’t too shabby, either: Neurovascular product sales were integral to the company-best gains experienced by Stryker’s Neurotechnology and Spine segment last year. Revenue climbed 5 percent (9.5 percent in constant currency) to $1.82 billion, driven largely by higher volume, product mix changes, and greater demand for neurotechnology devices, according to Stryker’s 2015 annual report.
That demand pumped up Neurotechnology proceeds 8.6 percent (14.2 percent in constant currency) to $1.08 billion—the second-largest product-specific increase within Stryker. Spinal sales, conversely, remained flat at $740 million, lagging behind the company’s broader portfolio due to pricing pressures, industry-wide efficacy concerns, and pure-play competition. The division could soon catch up to its brethren, however, as the company unveiled nearly a dozen new products at the North American Spine Society annual meeting last October. Potential cash cows include the Aero-LL, a lateral lumbar interbody and fixation system for use with autogenous bone graft in degenerative disc disease treatment; Tempus, an anterior cervical plating system featuring a secondary locking mechanism offering visual and tactile confirmation, large graft windows, and a low-profile design; BIO4, a next-generation viable bone matrix; BIOExpand demineralized cancellous sponge; and additions to the LITe (less invasive technologies) platform, including a decompression system and anterior and midline retractors, as well as anterior and lateral plates.

Stryker Corp. received U.S. Food and Drug Administration clearance last summer to use its Mako robotic surgery system for total knee construction procedures. (Courtesy of Stryker Corp.)
Stryker’s MedSurg segment welcomed some new additions as well in 2015. The Instruments division launched the 1588 AIM platform, a new camera system that enhances visualization of patient anatomy across six surgical specialties. The platform’s five key elements include IRIS (Infrared Illumination System), a visualization technology that aims to reduce the risk of ureteral damage; ENV (Endoscopic Near Infrared Visualization), designed to reduce the risk of common bile duct injury during laparoscopy; Clarity, intended to improve anatomy visualization by enhancing tissue level detail and intrabody structures, which can improve image quality up to 48 percent; DRE (Dynamic Range Enhancement), aimed at improving visualization in the surgical field by creating a brighter image in dark and posterior compartments; and Desaturation, the ability to decrease image color saturation.
“The reality is that this technology brings a lot to the table,” Santiago Horgan, M.D., chief of minimally invasive surgery at the University of California-San Diego, said when the 1588 system debuted in mid-December.
It brings a lot to imaging, really. But Stryker did literally bring some new technology to its line of hospital tables, beds, and patient furniture with its acquisitions of Turkish medical equipment manufacturer Muka Metal A.S. and Canadian low-weight hospital bed maker CHG Hospital Beds Inc. The Muka takeover significantly expanded Stryker’s presence in Turkey and Latin America, while the CHG purchase added to the company’s offerings an expandable low-height bariatric bed for acute care patients.
The purchases also boosted Medical revenue 7.5 percent (10.4 percent in constant currency) to $823 million. The accretion helped boost total MedSurg segment sales 3 percent (6.2 percent in constant currency) to $3.89 billion, though the unit’s overall performance also benefited from a 2.9 percent hike in Instruments proceeds ($1.46 billion) and a 3.4 percent increase in Sustainability revenue ($216 million). The gains, however, were slightly hampered by stagnating Endoscopy revenue, which crept up a mere $8 million to $1.39 billion.
Stryker’s Orthopaedics segment, which accounts for 43 percent of total company sales, posted the weakest growth in 2015 (year ended Dec. 31)—proceeds improved 1.7 percent (6.7 percent in constant currency) to $4.23 billion, driven mainly by a 13 percent surge in “Other” revenue ($266 million) and 4.9 percent jump in Trauma and Extremities sales ($1.29 billion). The increase, however, was offset by flatlining knee revenue ($1.4 billion) and a 2.1 percent decline in hip proceeds ($1.26 billion).
Hip sales remained hobbled by the company’s $1.43 billion Rejuvenate lawsuit settlement, announced in late 2014 and implemented last year. Stryker awarded victims of its faulty metal-on-metal Rejuvenate Modular Neck and ABG II Modular Neck implants roughly $300,000 each for the pain, problems, and injuries associated with the recalled hips. The majority of those payments were completed by the end of 2015.
Yet large joints weren’t a total drain on the Orthopaedics segment. The FDA gave the company permission to use its Mako robotic surgery system for total hip and knee replacements, expanding its indication for use with Stryker’s Triathlon Total Knee as well as the Accolade II, Secur-Fit Advanced, Anato, and Exeter femoral implants coupled with the Trident Acetabular system and engineered X3 polyethylene.
The broadened indications are a significant advance of Stryker’s strategy to differentiate itself through the Mako platform, which reportedly improves accuracy and surgical outcomes. The company already uses the Mako system to perform partial knee resurfacing.
Stryker is planning a limited market release of its new robotics offerings as it seeks to capitalize on the $1.65 billion acquisition of Mako Surgical in 2013. Adoption of the Mako system has been slower than expected due to concerns about the device’s cost and clinical benefits, but a four-prong approach to convince customers to invest in such a large capital investment (more than $1 million per surgical unit) has begun to pay off. The company sold 72 Mako units in 2015, with 43 percent of those systems (31) peddled in the fourth quarter alone.
The late-year surge in Mako sales helped lift Stryker’s 2015 revenue 2.8 percent to $9.94 billion. Gross profit rose 3.8 percent to $6.6 billion and net earnings nearly tripled, going from $515 million in 2014 to $1.43 billion last year, and net earnings per diluted share more than doubled, ending the year at $3.78.
Domestic sales outpaced the market, swelling 8.5 percent to $7.11 billion, but a second-half slowdown in China and Brazil contributed to a 9.2 percent decline in international sales ($2.83 billion). The trouble in those markets, however, was offset by solid growth in Europe, Japan, and Australia. Management realigned the company’s European businesses under the new Trans-Atlantic Operating Model in an effort to improve oversight and bring foreign sales more in line with the domestic business. Additionally, European Union headquarters relocated to Amsterdam, which produced a permanent tax benefit of more than 2 percent.
KEY EXECUTIVES:
Kevin A. Lobo, Chairman and CEO
Lonny J. Carpenter, Group President, Global Quality and Business Operations
Glenn S. Boehnlein, VP, Chief Financial Officer
David K. Floyd, Group President, Orthopaedics
Timothy J. Scannell, Group President, MedSurg & Neurotechnology
J. Andrew Pierce, President, Endoscopy
Spencer S. Stiles, President, Instruments
William J. Huffnagle, President, Reconstructive
Xavier Berling, President, Trauma and Extremities
Mark H. Paul, President, Neurovascular
Bradford L. Saar, President, Medical
Bradley W. Paddock, President, Spine
Wayne D. Dahlberg, President, Performance Solutions
NUMBER OF EMPLOYEES: 27,000
HEADQUARTERS: Kalamazoo, Mich.
Beth Roche always considered herself a “mover.”
“I’m a person who likes to get up and get it done,” she told a Chicago Tribune reporter several years ago.
Imagine her level of frustration, then, as she struggled to recover from injuries she sustained in the 2013 Boston Marathon bombing. The Highland, Ind., mother and avid runner was among hundreds wounded in the terrorist attack, suffering a compound fracture of her left tibia and a shattered kneecap.
Roche was in Beantown on the day of the blast to watch her daughter Rebecca run the marathon. The first of two homemade bombs exploded shortly after Rebecca crossed the finish line, spraying a leg-level pattern of shrapnel that killed three spectators and peeled back the skin on Roche’s knee “like a sardine can,” leaving the bones exposed.
Oddly, Roche felt no pain. Panic ensued as bystanders stampeded toward a nearby sports store; the wail of emergency sirens filled the air, mixing with piercing screams from the injured and frightened. “Run!” someone yelled to Roche.
“I can’t run,” she thought, unable even to stand.
Within minutes, public safety and rescue personnel covered Roche with a blanket and treated her bleeding wound with a tourniquet. Paramedics rushed her to Tufts Medical Center, where orthopedic trauma surgeons cleaned her wounds of debris (fishing out bits of the black canvas backpack that housed one of the bombs) and reconstructed her left knee in a four-hour surgery.
Although Roche lost a significant amount of skin, muscle, bone, and tendon in the attack, blood flowed to her lower leg and foot, enabling surgeons to save the limb. Dr. Scott Ryan, orthopedic trauma chief at Tufts, stabilized Roche’s damaged bones with rods and screws, and held her newly repaired (but still fragile) knee firmly in place with an external frame from Stryker Corp.
Composed of lightweight carbon-based materials, the Hoffmann II MRI External Fixation System is designed to provide patient comfort and facilitate fracture management and stabilization.
Cleared for magnetic resonance imaging use under prescribed conditions, the Hoffmann frame employs “snap-fit” technology, allowing doctors to conduct postoperative corrections and treatments with the device in place. Clinicians also can build the frame to treat fractures close to a joint (as in Roche’s case) or stay clear of damaged soft-tissue areas.
After undergoing three surgeries to remove scar tissue and replace lost ligaments, as well as months of grueling physical therapy, Roche has finally regained flexibility in her knee. She’s exceeded her doctors’ expectations for a modest recovery, reclaiming the ability to bend her knee 145 degrees (Ryan had predicted 90-degree flexibility). But chronic pain forces Roche to rethink stairs and occasionally doubt her ability to resume an active lifestyle.
Nevertheless, she refuses to give up hope.
“My life is a race now,” Roche said last fall. “It’s a physical, mental race to get back to normal. What my normal was before the bomb.”
Nora Kasapligil, 21, has already won that race. Her life returned to normal remarkably quickly—within a day of suffering an ischemic stroke in her Sonoma State University dorm room. Kasapligil awoke on June 5, 2015, with a severe headache, and after getting out of bed, walked straight into a wall. Luckily, her roommate noticed Kasapligil’s odd speech and movements and quickly summoned help, suspecting a stroke.
“I think I tried saying, ‘I have to go to work,’ and instead of it coming out like that, I said, ‘I have to go, go, go, go to work,’ ” the young woman recently recounted to a Northern California television station.
Medical scans at a local hospital eventually confirmed the roommate’s suspicions: a clot was blocking the blood supply to the right side of Kasapligil’s brain. But since it formed in her sleep, doctors weren’t sure they could use tissue plasminogen activator, or tPA, the U.S. Food and Drug Administration (FDA)-approved clot-busting drug that must be given intravenously with 4.5 hours to be effective. “After four and a half hours, the data tell us the risk of bleeding in the brain is greater than providing any benefit of opening the [blood] vessel,” explained Joey D. English, M.D., director of Neurointerventional Services at California Pacific Medical Center in San Francisco.
After confirming the affected brain tissue was still surviving, English and his colleagues treated Kasapligil’s stroke by removing her blood clot with a device developed by Stryker. The company’s Trevo XP ProVue Retriever clears blocked vessels using an expandable wire mesh stent that snares the clot and drags it out of the body (via catheters) through the femoral artery. The FDA-cleared device is billed as the world’s first stent retriever with full-length visibility, giving surgeons greater control and real-time information during placement, integration, and clot removal.
Within an hour of treatment with the Trevo XP device, Kasapligil’s left-side paralysis began to dissipate; her facial weakness and slurred speech disappeared a day later.
“The new stent retrieval devices for treating large-vessel occlusions are pretty remarkable advances,” English said. “I hate to think what Nora’s outcome would have been without this new technology. She’s living proof that Trevo has changed the way we treat stroke.”
Indeed, stent retrievers like the Trevo are rewriting the rules of ischemic stroke treatment. Last summer, the American Heart Association (AHA) and American Stroke Association issued guidelines recommending the use of stent retrievers in concert with tPA after five studies published in the New England Journal of Medicine found the devices reduced disability, improved neurological function, shortened recovery time, and increased the rate at which stroke survivors regain function. The guidelines advised the stent retrieval procedure be performed on adult patients within six hours of acute stroke symptoms, and only after imaging proves the affected cranial area is not permanently damaged. The endorsement was the AHA’s first in two decades.

The Trevo XP ProVue Retriever was one of the key products used in the clinical trials that prompted the American Heart Association and American Stroke Association to issue new guidelines last year recommending endovascular therapy with stent retrievers for ischemic stroke patients. (Courtesy of Stryker Corp.)
Though stent retrievers are available at more than 1,000 U.S. stroke centers and hospitals, only about 13,000 procedures are performed annually—mostly at comprehensive stroke centers—because they require specialized training. The devices are now so technically sophisticated that properly trained interventional neuroradiologists could regularly remove clots within an hour, with the best treatment time of just under 20 minutes from the initial computed tomography scan.
Despite their promise, however, retrievers like the ProVue will most likely remain on the periphery of standardized treatment without significant market evolution. “...an impressive amount of strong clinical data has been released supporting the use of device-based treatment of acute ischemic stroke,” Katherine Owen, Stryker’s vice president of strategy and investor relations, noted to analysts during a first-quarter earnings conference call last spring. “We have pioneered this space in our unique and differentiated products. We expect the market will take time to evolve, which will require the automation of EMS transport and inter-hospital transfers, establishment of clinical guidelines, position incentives, and investment in new human and physical capital to absorb new patient volume. Clearly as the data comes out, it reinforces our excitement from the ischemic stroke market and its longer-term revenue potential.”
The market’s short-term potential isn’t too shabby, either: Neurovascular product sales were integral to the company-best gains experienced by Stryker’s Neurotechnology and Spine segment last year. Revenue climbed 5 percent (9.5 percent in constant currency) to $1.82 billion, driven largely by higher volume, product mix changes, and greater demand for neurotechnology devices, according to Stryker’s 2015 annual report.
That demand pumped up Neurotechnology proceeds 8.6 percent (14.2 percent in constant currency) to $1.08 billion—the second-largest product-specific increase within Stryker. Spinal sales, conversely, remained flat at $740 million, lagging behind the company’s broader portfolio due to pricing pressures, industry-wide efficacy concerns, and pure-play competition. The division could soon catch up to its brethren, however, as the company unveiled nearly a dozen new products at the North American Spine Society annual meeting last October. Potential cash cows include the Aero-LL, a lateral lumbar interbody and fixation system for use with autogenous bone graft in degenerative disc disease treatment; Tempus, an anterior cervical plating system featuring a secondary locking mechanism offering visual and tactile confirmation, large graft windows, and a low-profile design; BIO4, a next-generation viable bone matrix; BIOExpand demineralized cancellous sponge; and additions to the LITe (less invasive technologies) platform, including a decompression system and anterior and midline retractors, as well as anterior and lateral plates.

Stryker Corp. received U.S. Food and Drug Administration clearance last summer to use its Mako robotic surgery system for total knee construction procedures. (Courtesy of Stryker Corp.)
“The reality is that this technology brings a lot to the table,” Santiago Horgan, M.D., chief of minimally invasive surgery at the University of California-San Diego, said when the 1588 system debuted in mid-December.
It brings a lot to imaging, really. But Stryker did literally bring some new technology to its line of hospital tables, beds, and patient furniture with its acquisitions of Turkish medical equipment manufacturer Muka Metal A.S. and Canadian low-weight hospital bed maker CHG Hospital Beds Inc. The Muka takeover significantly expanded Stryker’s presence in Turkey and Latin America, while the CHG purchase added to the company’s offerings an expandable low-height bariatric bed for acute care patients.
The purchases also boosted Medical revenue 7.5 percent (10.4 percent in constant currency) to $823 million. The accretion helped boost total MedSurg segment sales 3 percent (6.2 percent in constant currency) to $3.89 billion, though the unit’s overall performance also benefited from a 2.9 percent hike in Instruments proceeds ($1.46 billion) and a 3.4 percent increase in Sustainability revenue ($216 million). The gains, however, were slightly hampered by stagnating Endoscopy revenue, which crept up a mere $8 million to $1.39 billion.
Stryker’s Orthopaedics segment, which accounts for 43 percent of total company sales, posted the weakest growth in 2015 (year ended Dec. 31)—proceeds improved 1.7 percent (6.7 percent in constant currency) to $4.23 billion, driven mainly by a 13 percent surge in “Other” revenue ($266 million) and 4.9 percent jump in Trauma and Extremities sales ($1.29 billion). The increase, however, was offset by flatlining knee revenue ($1.4 billion) and a 2.1 percent decline in hip proceeds ($1.26 billion).
Hip sales remained hobbled by the company’s $1.43 billion Rejuvenate lawsuit settlement, announced in late 2014 and implemented last year. Stryker awarded victims of its faulty metal-on-metal Rejuvenate Modular Neck and ABG II Modular Neck implants roughly $300,000 each for the pain, problems, and injuries associated with the recalled hips. The majority of those payments were completed by the end of 2015.
Yet large joints weren’t a total drain on the Orthopaedics segment. The FDA gave the company permission to use its Mako robotic surgery system for total hip and knee replacements, expanding its indication for use with Stryker’s Triathlon Total Knee as well as the Accolade II, Secur-Fit Advanced, Anato, and Exeter femoral implants coupled with the Trident Acetabular system and engineered X3 polyethylene.
The broadened indications are a significant advance of Stryker’s strategy to differentiate itself through the Mako platform, which reportedly improves accuracy and surgical outcomes. The company already uses the Mako system to perform partial knee resurfacing.
Stryker is planning a limited market release of its new robotics offerings as it seeks to capitalize on the $1.65 billion acquisition of Mako Surgical in 2013. Adoption of the Mako system has been slower than expected due to concerns about the device’s cost and clinical benefits, but a four-prong approach to convince customers to invest in such a large capital investment (more than $1 million per surgical unit) has begun to pay off. The company sold 72 Mako units in 2015, with 43 percent of those systems (31) peddled in the fourth quarter alone.
The late-year surge in Mako sales helped lift Stryker’s 2015 revenue 2.8 percent to $9.94 billion. Gross profit rose 3.8 percent to $6.6 billion and net earnings nearly tripled, going from $515 million in 2014 to $1.43 billion last year, and net earnings per diluted share more than doubled, ending the year at $3.78.
Domestic sales outpaced the market, swelling 8.5 percent to $7.11 billion, but a second-half slowdown in China and Brazil contributed to a 9.2 percent decline in international sales ($2.83 billion). The trouble in those markets, however, was offset by solid growth in Europe, Japan, and Australia. Management realigned the company’s European businesses under the new Trans-Atlantic Operating Model in an effort to improve oversight and bring foreign sales more in line with the domestic business. Additionally, European Union headquarters relocated to Amsterdam, which produced a permanent tax benefit of more than 2 percent.