KEY EXECUTIVES:
Alex Gorsky, Chairman and CEO
Dominic J. Caruso, VP, Finance and Chief Financial Officer
Gary J. Pruden, Worldwide Chairman, Medical Devices
Sandra E. Peterson, Group Worldwide Chairman
Kathryn E. Wengel, VP, Johnson and Johnson Supply Chain
Michael E. Sneed, VP, Global Corporate Affairs
Ronald A. Kapusta, Corporate Controller, Chief Accounting Officer
NUMBER OF EMPLOYEES: 23,000
HEADQUARTERS: Warsaw, Ind.; Raynham, Mass.; and West Chester, Pa.
“Innovation is the specific instrument of entrepreneurship. The act that endows resources with a new capacity to create wealth.”
— Peter Drucker
Innovation is a woefully misunderstood concept. Ubiquitous, mysterious, and stubbornly elusive, that darling of business buzzwords has become synonymous with the loftiest of human ambitions: power, fame, and wealth. Like its counterpart, “leadership,” the word can stand firmly on its own but also pairs well with many adjectives; it signifies both everything and nothing, assuming various meanings depending on interpretation.
Yet despite its popularity (Amazon lists more than 70,000 book titles on the subject), innovation is, at best, a vague, loosely-defined concept within most corporate circles. Its basic definition—systematically finding, encouraging, and implementing new ideas—has become buried beneath all the variations, nuances, tools, techniques, models, frameworks, and paradigms associated with the word.
Even in its most elemental form, though, innovation isn’t quite so simple. It’s certainly not as basal as the old adage alleges—necessity is not always the sole spawn of invention. “We can all agree that a problem can be a catalyst for a solution, and that many business innovations are born of business challenges,” author/speaker/business strategist/consultant/entrepreneur Larry Myler wrote in a June 2014 Forbes column. “But is it really that simple?”
Not really. Myler contends there are four distinct levels of innovation, the first being problem-solving. Deemed a “reactive” approach to innovation, this method can be both powerful and prolific as long as the problems and solutions are chosen and crafted carefully, Myler claims.
Preventing problems (Level No. 2) is an effective approach as well, though it can be difficult to find and rally the proper resources for executing change. Likewise, “continuous improvement” (Level No. 3) helps companies evolve with the market, but it also can endanger their past progress. “If it ain’t broke, break it is a fun maxim...” Myler writes in his column, “but...replacing a perfectly good business system, albeit for the greater good in the end, creates real problems in the near-term.”
Or, it can act as a springboard to the final level (four), where innovation is achieved through reinvention, or “creation of a new future.” This method, however, is one of the most challenging ways to innovate, as it requires companies abandon traditional ideation discovery processes in favor of unconventional practices. The Henry Ford Innovation Institute and companies like Google and Facebook, for instance, are banking their futures on impromptu workforce “collisions” designed to encourage random (but slightly orchestrated) encounters and idea-sharing among employees. Similarly, Yahoo banned telecommuting in 2013 to boost its grassroots thinktank prowess.
Such forward-thinking strategies, of course, are not foolproof, but without them companies risk possible extinction. “With today’s unprecedented pace of change, the pressure on companies to reinvent themselves is relentless,” Myler noted. “It is sobering to consider the number of companies...that have fallen into mediocrity or worse. Disruptive forces are constantly lurking in the shadows, ready to pounce on unwary, and therefore unprepared, successful companies. Staying relevant and competitive is no easy task.”
The job can be particularly difficult for large multinationals, which often fall victim to their own complacency, inflexibility, and tunnel vision. Johnson & Johnson currently is struggling with professional relevancy as it wrestles with a medtech market slowdown driven by cost-conscious hospitals and group purchasing organizations.
The $381 billion global market for medical devices is growing 3 to 4 percent annually, down from a double-digit rate in the early part of the millennium, according to various industry reports. JNJ’s medical device sales have slowed dramatically in the last few years, falling 7.3 percent and 3.3 percent respectively, in the final two quarters of 2015.
Despite the downturn, the 130-year-old company is confident it can remain a formidable force in the medical device sector by tackling innovation on several different levels.
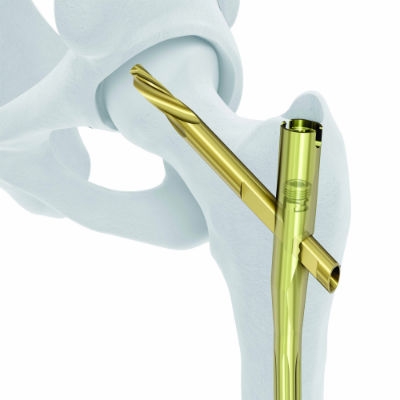
DePuy Synthes launched the TFN-Advanced Proximal Femoral Nailing System at the American Academy of Orthopaedic Surgeons 2015 Annual Meeting. The product is designed to improve patient outcomes by more closely matching the anatomy of patients undergoing hip fracture surgery. (Courtesy of DePuy Synthes)
“For 130 years, our company has been driving breakthrough innovation in health care—from revolutionizing wound care in the 1880s to developing cures, vaccines, and treatments for some of today’s most pressing diseases in the world,” CEO and Board Chairman Alex Gorsky wrote in his 2015 annual letter to shareholders. “We are acutely aware of the need to evaluate our business against the changing health care environment and to challenge ourselves based on the results we deliver. At Johnson & Johnson, we believe the most important contribution we can make to the dynamic challenges we are facing is innovation—innovation in products, services, solutions and in everything we do.”
Some of those solutions have been crafted using Myler’s second-level approach to innovation (i.e., problem prevention). Over the last few years, the company has steadily been streamlining its Medical Device division, a former pecuniary powerhouse that once generated significant profits from its sales of stents, artificial joints, and surgical tools. But pricing pressure and strong competition from Medtronic plc, Abbott Laboratories, Boston Scientific Corp., and lesser-known firms has gradually eroded JNJ’s market share.
Consequently, the company has all but abandoned the stent business it had pioneered, and has been selling small, slower-growing units. Last March, for example, JNJ sold its Cordis business to Cardinal Health for $1.9 billion in cash, and in June 2014, hawked its Ortho-Clinical Diagnostics unit to private equity firm The Carlyle Group for $4.2 billion. Yet even with those two transactions, the company still has 11 different medical device businesses, including BioSense Webster, the troubled orthopedic and neurological-focused DePuy Synthes, Janssen Diagnostics, and advanced surgical care units Ethicon and Ethicon Endo-Surgery.
“We have to put the patient in the center and reward innovations that drive better outcomes and long-term value,” Gorsky told investors at the start of JNJ’s 2015 annual report. “The promise of innovation in health care is great, but it comes with the need for forward-focused investment in R&D, a holistic approach to evolving global health care markets and bold future-facing strategies.”
Those strategies could potentially help JNJ reinvent itself, thereby helping the New Brunswick, N.J.-based company attain the fourth level of innovation. JNJ’s multi-faceted gameplan involves revamping its selling strategy, focusing on key markets (China, Japan, and the United States), infiltrating high-growth areas (e.g., staplers and surgical robotics), and collaborating to develop professional education programs as well as patient data collection and surgical care initiatives.
The company’s partnership with International Business Machines Corp. (IBM)—announced last spring—will leverage the computing giant’s Watson cognitive computation and analytics platform to create a personal concierge service that prepares hip and knee replacement candidates for surgery and post-operative care. Similarly, JNJ’s union with the AO Foundation is designed to deliver professional education and develop innovations to improve both patient outcomes and efficiency of care, a journey both entities jointly began in 1960. The two organizations are collaborating on new innovation and will work to certify DePuy Synthes products by the AO Technical Commission for use in the foundation’s educational activities, in which more than 200,000 healthcare professionals are expected to participate during the next five years.
JNJ is also forging a new future through M&A. Its DePuy Synthes division, for example, added new capabilities to its lineup with the purchase of Salt Lake City, Utah-based Olive Medical Corporation in February 2015. Founded in 2009, Olive Medical manufactures high-definition visualization systems for minimally invasive surgery; its camera control unit and camera head capture full HD video, and illuminate consistent white light to the surgical site.
The deal gives DePuy Synthes Mitek Sports Medicine full use of Olive Medical’s imaging technology in its arthroscopy product line, presumably improving visualization in patients with shoulder, hip, knee, and small-joint maladies. It could also help JNJ simplify supplier networks for clinicians and medical service providers, company officials noted.
“Because of our broad base across healthcare, we are uniquely positioned to be a partner of choice,” Gorsky said during a 2015 earnings conference call earlier this year. “We’re collaborating more than ever before to meet the world’s most pressing public health challenges…being broadly based in healthcare has been and will continue to be a significant factor in Johnson & Johnson’s success, as it positions us well to lead and grow our business with the evolving healthcare landscape.”
Most of the time, anyway. Last year, JNJ’s broad-base growth strategy encountered a setback, as sales were down in each of the company’s three business segments. The Medical Devices segment posted the most sizable loss, resulting mostly from a 1.4 percent operational decline, a 7.3 percent negative currency impact, and divestitures of the Ortho-Clinical Diagnostics and Cordis businesses—all of which contributed to a $2.38 billion decrease in the unit’s 2015 total revenue. U.S. sales slipped 1 percent to $12.1 billion, while international proceeds fell 14.8 percent to $13 billion due to a 1.7 percent operational decrease and a 13.1 percent negative currency impact.
The Medical Devices segment’s losses were sweeping: Deficits occurred in all six franchises and six of the seven divisions (only Advanced Surgery escaped with a 1.2 percent improvement on its 2014 performance). The Orthopaedics franchise fared best in the carnage, keeping its year-over-year shortfall to 4.3 percent, or half of JNJ’s 2015 total sales descent. Proceeds declined by $413 million amid soft product demand, pricing pressures, and inventory level reductions (mostly in China), according to annual report data.
Orthopaedics did, however, find some traction in the Attune Knee System, trauma TFNA nailing system, and Orthovisc/Monovisc sports medicine products.

DePuy Synthes touts its Conform Flex with Q-PACK technology as the only pre-hydrated demineralized cancellous bone tissue matrix designed for use in trauma-related extremity procedures, including fusion, and for filling bone voids. (Courtesy of DePuy Synthes)
Despite the growth in Attune sales, total knee revenue fell 2.4 percent to $1.49 billion. Similarly, the March launch of the TFN-Advanced Proximal Femoral Nailing System and October debut of the Conform Flex pre-hydrated bone biologic did little to stop the 4.2 percent slide in 2015 trauma proceeds, which totaled $2.52 billion.
Unveiled at the 2015 Orthopaedic Trauma Association Annual Meeting, the Conform Flex with Q-Pack technology was developed specifically for trauma-related extremity cases (foot, ankle, hand, wrist), including fusion, and for filling bone voids. Both osteoconductive (provides a natural scaffold for new bone growth) and osteoinductive (stimulates new bone formation), the biologic implant remains stationary once it is implanted; readily absorbs and retains bone aspirate, blood, and saline; and can help accelerate bone growth when mixed with bone marrow aspirate. The implant is also highly flexible and can be compressed up to 50 percent of its original size.
“What I appreciated about Conform Flex was the ease of use and its ability to conform easily in the joints that I had prepared for fusion,” Stephen Carley, M.D., with Mary Washington Hospital in Fredericksburg, Va., noted in a news release. “I was especially impressed by the way it maintained its position and integrity during positioning and compression of the fusion sites with internal fixation.”
Christopher Finkemeier, M.D., of Roseville, Calif., was equally as impressed by the TFNA nail, a sturdy titanium alloy product intended to better fit the anatomy of hip fracture patients. Curved to help surgeons avoid impingement in the lower inside part of the femur, the nail features a lateral relief cut and small diameter at the top to better preserve bone and ensure a proper fit, according to JNJ. The nail also comes equipped with a Helical blade that secures it to the femur head, allowing for proper healing, as well as a design that compresses bone around the implant during insertion, improving implant fixation.
“A big concern, especially in elderly patients with hip fractures, is matching patient anatomy to help avoid complications such as anterior cortical impingement, and the radius of curvature of the TFNA Nail may help reduce that risk,” Finkemeier, co-director of the Acute Care Orthopaedic Service at Sutter Roseville Medical Center (Roseville, Calif.) and member of the AO TK Intramedullary Nailing Expert Group, said upon the product’s release. “Having one system with both a screw and blade and simplified instrumentation are important for the surgeon as well. This system may help improve outcomes and minimize complications, with less time and cost in the O.R.”
The VAPR radiofrequency (RF) devices released by Mitek Sports Medicine last summer are designed to improve outcomes as well. The VAPR Tripolar 90 Suction Electrode—made for shoulder arthroscopy—is touted by the company as the first RF device with a proprietary tip design that independently optimizes ablation and coagulation performance. The product incorporates integrated suction control to help surgeons better visualize and target tissue for resection or coagulation, according to Mitek.
The VAPR Arctic Suction Electrode, on the other hand, is designed for hip arthroscopy. It combines bipolar energy flow, integrated suction and the ability to flex up to 110 degrees—features that help eliminate the need for additional saline suction ports or use of multiple electrodes during the same procedure, while providing access to previously hard-to-reach areas of the hip. The device also enhances operational efficiency: A study conducted by Mitek showed the Arctic Suction Electrode ablated 216 percent more soft tissue per minute than other RF electrodes.
While impressive, the Arctic’s exemplary ablation rate couldn’t rouse JNJ’s sagging hip sales. Revenue slipped 2.6 percent last year to its 2013 total of $1.33 billion.
Likewise, DePuy Synthes’ new spinal products were virtually useless in lifting Spine & Other proceeds in 2015 (year ended Dec. 31). Sales in that division took the biggest franchise tumble, falling 5.5 percent to $3.9 billion despite the introduction of the Expedium Verse Spinal system, described as an “all-in-one” pedicle screw system; the Expedium Osteotomy System, an instrument set for complex deformity correction surgeries; the Viper Cortical Fix X-Tab screw, designed for enhanced fixation in percutaneous surgery; the Synapse System, which uses 3.5-mm and 4-mm titanium alloy rods; and the Vivigen Cellular Bone Matrix, designed for use in the repair or reconstruction of musculoskeletal defects and for enhancements to the Synapse System of implants and instruments, enabling more surgical options during upper spine stabilization procedures.
Other unsuccessful (but still valiant) sales-boosting efforts involved the company’s newest platform of embolic coils for brain aneurysm treatment—MicrusFrame Coils for framing, Galaxy G3 Coils for filling, and DeltaXSFT Coils for finishing—as well as the MatrixWAVE MMF System, an adaptable, wave-patterned bone-borne plate and screw for the stabilization of mandibular and maxillary fractures and osteotomies.
The MatrixWAVE comprises short and tall plates with a wave pattern that allows surgeons to refine bone segment alignment after wiring without requiring them to reposition screws. The plates—which attach to the lower and upper jaw with 1.85-mm diameter screws with rounded heads—can be stretched or compressed for positioning away from tooth roots and nerves to help reduce the risk of soft tissue injury.