08.09.16
$545 Million
KEY EXECUTIVES:
David C. Paul, Chairman and CEO
Anthony L. Williams, President
Daniel T. Scavilla, Sr. VP and CFO
David M. Demski, Group President, Emerging Technologies
A. Brett Murphy, Group President, Commercial Operations
NUMBER OF EMPLOYEES: 1,200
HEADQUARTERS: Audubon, Pa.
During its 2015 fiscal year, Globus Medical enjoyed a relatively quiet period while seeing sales increase by double-digit figures over the prior year. Unfortunately, the good news of a positive financial year did not last very long. Just a couple of weeks following the year’s close, a lawsuit between Globus Medical and DePuy Synthes was settled for the amount of $7.9 million. The settlement covered several, originally separate lawsuits between the two companies—one filed by N-Spine and Synthes USA in 2010, a second by Synthes USA in 2011, and a third filed by DePuy Synthes in 2014. Both the 2010 and 2014 lawsuits alleged the infringement of a patent by Globus Medical for its Transition stabilization system product. The 2011 lawsuit alleged infringement against a patent by Globus with its Coalition, Independence, and Intercontinental products. The settlement of Jan. 13, 2016, brought to an end a total of four pending lawsuits between Globus Medical and DePuy Synthes.
Unfortunately, this issue wasn’t the only legal affair for Globus Medical in 2015. In a lawsuit dating back to March 2012 that had Sabatino Bianco filing suit against Globus for misappropriated trade secrets and confidential information by improperly utilizing it in the Caliber, Caliber-L, and Rise products, an appeals court affirmed the original judgment in favor of Bianco on Oct. 19, 2015. That decision goes back to a jury verdict in favor of Bianco on the claim of misappropriation of trade secret that was handed down in January 2014. A later court awarded Bianco an ongoing royalty of 5 percent on the net sales of all three products for a period of 15 years. Globus is still considering appeal options.
Two additional lawsuits were filed against Globus in 2015. Flexuspine Inc. filed for patent infringement against Globus, alleging that the company’s Caliber, Caliber-L, Rise, Rise-L, Intralif, and Altera products infringe on five of Flexuspine’s patents. The second lawsuit involves a putative securities class action lawsuit filed against Globus. The plaintiff purports to represent a class of stockholders who purchased shares in the company between Feb. 26, 2014, and Aug. 5, 2014. In both cases, the plaintiffs are seeking an unspecified amount plus relief. Globus is currently fighting both cases and offers no estimate as to when either will be resolved.
Legal concerns aside, Globus celebrated growth in sales in 2015. Its $545 million sales figure represented a 14.8 percent increase over 2014’s $474 million figure. The company’s sales are driven by two major product categories—Innovative Fusion and Disruptive Technologies. The Innovative Fusion group includes products that are used to “treat a wide variety of spinal disorders for the entire spine and can be used in a variety of surgical approaches,” according to the company. Globus views this assortment of products as presenting advantages to surgeons and patients, contributing to better outcomes. This group contributed $288 million to the total 2015 sales figure, which marked an increase of 6.4 percent over the prior year.
Globus’ other sector, Disruptive Technologies, are classified as products that potentially offer a “significant shift in the treatment of spinal disorders by allowing for novel surgical procedures, improvements to existing surgical procedures, and the treatment of spinal disorders earlier in the continuum of care,” according to the company. These products enable minimally invasive procedures, use of motion preservation technologies, and the employment of interventional pain management solutions. The product group is viewed by the company as a significant opportunity for growth. That strategy was supported by the overall impact of the group on the company’s sales. It represented approximately $257 million in sales toward the full 2015 figure, a significant 26.1 percent increase over 2014 (the group only saw a 12.8 percent increase the year prior).
Further, the company will likely see both sectors continue to grow with a healthy investment in research and development expenditures. The company put 6.8 percent of sales toward R&D in 2015, a figure that was 16.7 percent higher in actual dollar total over 2014. The company will likely see further growth in R&D in 2016 as it plans to redirect approximately 40 percent of money previously allocated to pay for the device tax that it had been responsible for in 2014 and 2015 (when the company paid $7.1 million and $8.1 million respectively) toward job-creation initiatives in R&D and manufacturing.
Additional growth opportunities lie in expansion of sales outside the United States, where company executives see an overwhelming opportunity. The United States represents almost $500 million of the company’s total sales, leaving the rest of the world as making up just $47 million of total sales or about 9 percent. The U.S. sales total represented an increase of 16.6 percent over 2014. While on a constant currency basis, international sales actually improved by 16 percent, the actual figure marked a decrease of 1.5 percent over the prior year.
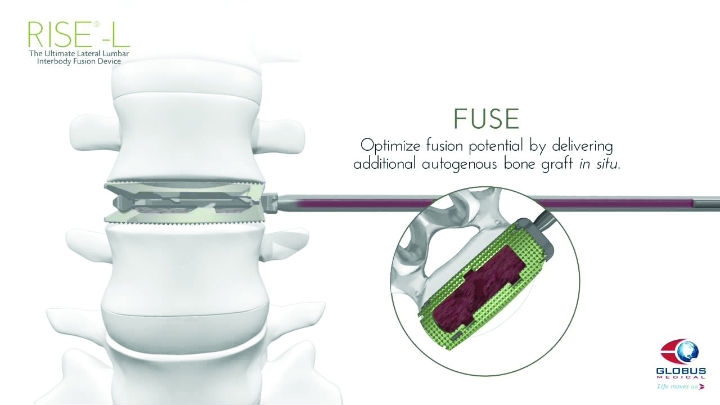
The Globus Medical Rise-L technology, released in July 2015. (Courtesy of Globus Medical)
In addition to working with international distributors to better identify market opportunities, Globus plans to continue to use product launches to help further grow international sales (in addition to the U.S. marketplace). The company saw a number of product releases in 2015, but had two that were notable.
In July, Globus announced the introduction of the Rise-L, an expandable lateral lumbar interbody fusion (LLIF) device. The product joined a line of expandable spacers the company already offered that was started with the Patriot TransContinetal LLIF cage.
“Over the last four years, we have continued to build upon our unmatched family of expandable spacers, and I am proud of our product development team with the launch of Rise-L. This new lateral cage eliminates the need to forcefully impact the cage into the disc space. Our expansion technology allows the Rise-L cage to be placed in compact form, and expanded for optimal fit, and also includes a large port for easy graft delivery once expanded,” Andrew Iott, senior vice president of product development, commented in the product release announcement.
Later in the year, in October, the second product release of note was announced—the Creo MIS, a low profile minimally invasive posterior stabilization system. The product featured one of the smallest screw head diameters available on the market at the time.
“Globus continues to lead the way with a growing portfolio of MIS related products, including this important addition to our Creo Pedicle Screw System platform. Surgeons appreciate the intuitive simplicity of the Creo MIS platform, including the ability to insert the locking cap through the screw head in a controlled, guided manner where the rod is then captured, reduced, and locked in one simplified step,” Iott commented in a company release. “Creo MIS has been used in over 300 spine procedures since its launch in the second quarter of 2015, and the feedback from surgeons has been extremely positive. We are proud to add yet another innovative solution to our suite of MIS products. This product is designed to help surgeons treat their patients with less tissue disruption leading to faster recovery times. We will continue to provide innovative solutions that allow our surgeons to better improve the lives of their patients.”
The company may also enjoy growth opportunities through its acquisition in 2015 of Branch Medical Group (BMG) for the purchase price of $52.9 million. The company was a third party manufacturer of high-precision medical devices.
“We are very excited by the vertical integration opportunity afforded by BMG to strengthen Globus, both operationally and financially. BMG is an extremely efficient organization; driven by their “Right, On Time” credo to produce high-quality, precision medical devices,” Dave Demski, president and COO of Globus, said in a press release put out to announce the acquisition. “The potential to expand the amount of products manufactured by BMG in the future represents a significant opportunity for Globus, and, at the acquisition price, is a compelling value.”
The established relationship between Globus and BMG was also viewed as a significant advantage that would only increase the value of the acquisition.
“BMG has grown to be our top supplier by consistently producing our most complex parts in a highly efficient manner. The close working relationship we have enjoyed over the years has enabled us to work together to get products to the market quickly,” Iott said in the same release. “The ability for enhanced collaboration between Globus engineers and BMG manufacturing professionals in the future is an added benefit to the transaction.”
KEY EXECUTIVES:
David C. Paul, Chairman and CEO
Anthony L. Williams, President
Daniel T. Scavilla, Sr. VP and CFO
David M. Demski, Group President, Emerging Technologies
A. Brett Murphy, Group President, Commercial Operations
NUMBER OF EMPLOYEES: 1,200
HEADQUARTERS: Audubon, Pa.
During its 2015 fiscal year, Globus Medical enjoyed a relatively quiet period while seeing sales increase by double-digit figures over the prior year. Unfortunately, the good news of a positive financial year did not last very long. Just a couple of weeks following the year’s close, a lawsuit between Globus Medical and DePuy Synthes was settled for the amount of $7.9 million. The settlement covered several, originally separate lawsuits between the two companies—one filed by N-Spine and Synthes USA in 2010, a second by Synthes USA in 2011, and a third filed by DePuy Synthes in 2014. Both the 2010 and 2014 lawsuits alleged the infringement of a patent by Globus Medical for its Transition stabilization system product. The 2011 lawsuit alleged infringement against a patent by Globus with its Coalition, Independence, and Intercontinental products. The settlement of Jan. 13, 2016, brought to an end a total of four pending lawsuits between Globus Medical and DePuy Synthes.
Unfortunately, this issue wasn’t the only legal affair for Globus Medical in 2015. In a lawsuit dating back to March 2012 that had Sabatino Bianco filing suit against Globus for misappropriated trade secrets and confidential information by improperly utilizing it in the Caliber, Caliber-L, and Rise products, an appeals court affirmed the original judgment in favor of Bianco on Oct. 19, 2015. That decision goes back to a jury verdict in favor of Bianco on the claim of misappropriation of trade secret that was handed down in January 2014. A later court awarded Bianco an ongoing royalty of 5 percent on the net sales of all three products for a period of 15 years. Globus is still considering appeal options.
Two additional lawsuits were filed against Globus in 2015. Flexuspine Inc. filed for patent infringement against Globus, alleging that the company’s Caliber, Caliber-L, Rise, Rise-L, Intralif, and Altera products infringe on five of Flexuspine’s patents. The second lawsuit involves a putative securities class action lawsuit filed against Globus. The plaintiff purports to represent a class of stockholders who purchased shares in the company between Feb. 26, 2014, and Aug. 5, 2014. In both cases, the plaintiffs are seeking an unspecified amount plus relief. Globus is currently fighting both cases and offers no estimate as to when either will be resolved.
Legal concerns aside, Globus celebrated growth in sales in 2015. Its $545 million sales figure represented a 14.8 percent increase over 2014’s $474 million figure. The company’s sales are driven by two major product categories—Innovative Fusion and Disruptive Technologies. The Innovative Fusion group includes products that are used to “treat a wide variety of spinal disorders for the entire spine and can be used in a variety of surgical approaches,” according to the company. Globus views this assortment of products as presenting advantages to surgeons and patients, contributing to better outcomes. This group contributed $288 million to the total 2015 sales figure, which marked an increase of 6.4 percent over the prior year.
Globus’ other sector, Disruptive Technologies, are classified as products that potentially offer a “significant shift in the treatment of spinal disorders by allowing for novel surgical procedures, improvements to existing surgical procedures, and the treatment of spinal disorders earlier in the continuum of care,” according to the company. These products enable minimally invasive procedures, use of motion preservation technologies, and the employment of interventional pain management solutions. The product group is viewed by the company as a significant opportunity for growth. That strategy was supported by the overall impact of the group on the company’s sales. It represented approximately $257 million in sales toward the full 2015 figure, a significant 26.1 percent increase over 2014 (the group only saw a 12.8 percent increase the year prior).
Further, the company will likely see both sectors continue to grow with a healthy investment in research and development expenditures. The company put 6.8 percent of sales toward R&D in 2015, a figure that was 16.7 percent higher in actual dollar total over 2014. The company will likely see further growth in R&D in 2016 as it plans to redirect approximately 40 percent of money previously allocated to pay for the device tax that it had been responsible for in 2014 and 2015 (when the company paid $7.1 million and $8.1 million respectively) toward job-creation initiatives in R&D and manufacturing.
Additional growth opportunities lie in expansion of sales outside the United States, where company executives see an overwhelming opportunity. The United States represents almost $500 million of the company’s total sales, leaving the rest of the world as making up just $47 million of total sales or about 9 percent. The U.S. sales total represented an increase of 16.6 percent over 2014. While on a constant currency basis, international sales actually improved by 16 percent, the actual figure marked a decrease of 1.5 percent over the prior year.

The Globus Medical Rise-L technology, released in July 2015. (Courtesy of Globus Medical)
In addition to working with international distributors to better identify market opportunities, Globus plans to continue to use product launches to help further grow international sales (in addition to the U.S. marketplace). The company saw a number of product releases in 2015, but had two that were notable.
In July, Globus announced the introduction of the Rise-L, an expandable lateral lumbar interbody fusion (LLIF) device. The product joined a line of expandable spacers the company already offered that was started with the Patriot TransContinetal LLIF cage.
“Over the last four years, we have continued to build upon our unmatched family of expandable spacers, and I am proud of our product development team with the launch of Rise-L. This new lateral cage eliminates the need to forcefully impact the cage into the disc space. Our expansion technology allows the Rise-L cage to be placed in compact form, and expanded for optimal fit, and also includes a large port for easy graft delivery once expanded,” Andrew Iott, senior vice president of product development, commented in the product release announcement.
Later in the year, in October, the second product release of note was announced—the Creo MIS, a low profile minimally invasive posterior stabilization system. The product featured one of the smallest screw head diameters available on the market at the time.
“Globus continues to lead the way with a growing portfolio of MIS related products, including this important addition to our Creo Pedicle Screw System platform. Surgeons appreciate the intuitive simplicity of the Creo MIS platform, including the ability to insert the locking cap through the screw head in a controlled, guided manner where the rod is then captured, reduced, and locked in one simplified step,” Iott commented in a company release. “Creo MIS has been used in over 300 spine procedures since its launch in the second quarter of 2015, and the feedback from surgeons has been extremely positive. We are proud to add yet another innovative solution to our suite of MIS products. This product is designed to help surgeons treat their patients with less tissue disruption leading to faster recovery times. We will continue to provide innovative solutions that allow our surgeons to better improve the lives of their patients.”
The company may also enjoy growth opportunities through its acquisition in 2015 of Branch Medical Group (BMG) for the purchase price of $52.9 million. The company was a third party manufacturer of high-precision medical devices.
“We are very excited by the vertical integration opportunity afforded by BMG to strengthen Globus, both operationally and financially. BMG is an extremely efficient organization; driven by their “Right, On Time” credo to produce high-quality, precision medical devices,” Dave Demski, president and COO of Globus, said in a press release put out to announce the acquisition. “The potential to expand the amount of products manufactured by BMG in the future represents a significant opportunity for Globus, and, at the acquisition price, is a compelling value.”
The established relationship between Globus and BMG was also viewed as a significant advantage that would only increase the value of the acquisition.
“BMG has grown to be our top supplier by consistently producing our most complex parts in a highly efficient manner. The close working relationship we have enjoyed over the years has enabled us to work together to get products to the market quickly,” Iott said in the same release. “The ability for enhanced collaboration between Globus engineers and BMG manufacturing professionals in the future is an added benefit to the transaction.”






















