08.15.17
$690 Million
KEY EXECUTIVES:
Robert J. Palmisano, President and CEO
Lance A. Berry, Sr. VP and CFO
Robert P. Burrows, Sr. VP, Supply Chain
J. Wesley Porter, Sr. VP and Chief Compliance Officer
Julie D. Tracy, Sr. VP and Chief Communications Officer
Jennifer S. Walker, Sr. VP, Process Improvement
Kevin D. Cordell, President, U.S.
Peter S. Cooke, President, International
Timothy L. Lanier, President, Upper Extremities
Patrick Fisher, President, Lower Extremities
NO. OF EMPLOYEES: 2,394
GLOBAL HEADQUARTERS: Amsterdam, the Netherlands
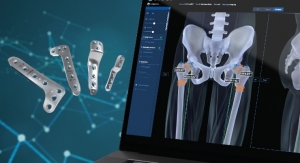
Extremities and biologics—two of fastest growing areas of orthopedic technologies. Not coincidentally, these same two areas have become the primary focus for Wright Medical Group. Over the last several years following a series of strategic transactions, the company has positioned itself to focus almost exclusively on these two market sectors.
Beginning in early 2014, Wright Medical divested its hip and knee business (i.e., OrthoRecon) to MicroPort Scientific Corporation. It followed that up with a substantial merger with a leading orthopedic OEM serving the extremities and biologics space—Tornier—which closed Oct. 1, 2015. In 2016 (the fiscal year closed Dec. 25), the company rapidly realized the synergies of that merger, having accomplished more than 80 percent of its approximately 300 integration milestones during the year.
Still seeking to further sharpen its pinpoint focus on the two markets, in July 2016, Wright announced that it was parting ways with its large joints business. Corin Orthopaedics Holdings Ltd. paid approximately $33 million for the hips and knees business. The legacy Tornier unit had become a portion of Wright following the merger. The business sold knee and hip implants primarily in France and other European countries with a product line that included Dynacup and Meije Duo hip implants, as well as HLS KneeTec and HLS Noetos knee implants.
“Our large joints business has excellent products and significant market share in key European markets with a loyal customer base. However, this business is not in line with our strategy to be the premier extremities and biologics company,” Robert Palmisano, president and CEO of Wright Medical, said in a statement regarding the sale. “Post-closing, we will be able to devote our full resources and attention on accelerating growth opportunities in the high-growth extremities and biologics markets and believe this will enhance our ability to create significant shareholder value.”
That focus even permeated the company’s 2016 annual report, with Palmisano kicking it off in his letter with a single-minded message to shareholders, customers, and employees: “Our Focused Excellence approach has enabled us to transform our company to become the leader in the fastest-growing orthopedics markets: upper extremities, lower extremities, and biologics.”
The strategy’s success could be readily observed in the company’s financials for FY16. In the United States (the OEM’s overwhelmingly strongest commercial market), it saw gains in both Upper and Lower Extremities businesses of 14.3 percent and 7.6 percent respectively, over the firm’s 2015 sales in these categories. More impressive was the sales increases seen in the Biologics business. Experiencing a 43.6 percent increase over 2015, the business unit contributed $75 million to Wright’s $690 million total for the year. The company’s fourth business unit—Sports Medicine & Other—remained flat compared to the year prior with $8.4 million in sales. This division offers products used across several anatomic sites to mechanically repair tissue-to-tissue or tissue-to-bone injuries. In many cases, the products complement the upper and lower extremities product portfolios. Overall, the U.S. sales experienced an increase of 14.3 percent over year prior to finish at $507 million.
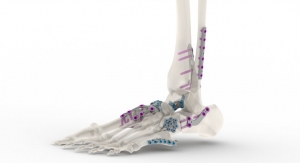
Primarily responsible for driving the remarkable growth seen in the U.S. Biologics unit was the launch of Wright’s Augment Bone Graft, which was commercially launched in the fourth quarter of 2015. Augment is an alternative to autograft for ankle and/or hindfoot fusion indications, saving the patient and surgeon the time and healing involved with an additional procedure that would be required to harvest the tissue. The biologic product, which was acquired from BioMimetic Therapeutics Inc. in March 2013, had previously faced a rejection from the U.S. Food and Drug Administration (FDA) in 2013 in response to the company’s PMA submission. Two years later, the technology gained the necessary approval and was launched.
“The FDA approval of Augment marks a capstone achievement that demonstrates the strength of our science and provides a breakthrough therapeutic option as an alternative to autograft in ankle and hindfoot fusion procedures,” said Palmisano. “This approval also underscores the significant effort and perseverance from our clinical trial investigators and Wright’s clinical, regulatory, and legal teams to bring the product to market. This success can be attributed to everyone involved in the clinical trial, especially the study investigators and coordinators; our R&D, regulatory, and clinical teams; our partners and suppliers; and most importantly, the patients who participated in the landmark prospective, randomized study comparing Augment Bone Graft to autograft.”
While the company’s international sales aren’t as substantial as those in the United States, they still represent a significant portion of the company’s overall sales for the year. Growth, unfortunately, was not as optimistic as the U.S. sales either, but overall, international still saw a 6.6 percent gain over 2015 to total $183 million. That number breaks down in a similar manner to the U.S. splits, with Lower Extremities ($63 million) and Upper Extremities ($87 million) representing the bulk of the company’s business. Those sales totals represent a 6.7 percent increase for Lower Extremities over 2015, while Upper Extremities experienced the greatest growth among international sales between the business units at 11.8 percent compared to FY15. Biologics ($19 million) and Sports Medicine & Other ($15 million), on the other hand, both saw sales decline in international markets by 5.7 percent and 4.1 percent respectively, compared to the prior year.
Before wrapping up what had amounted to a relatively successful year in terms of market growth and sales increases, new product launches, and gained efficiencies from the 2015 Tornier merger, Wright Medical resolved some pending legal issues with regard to its wholly owned subsidiary Wright Medical Technology.
The company entered into a Master Settlement Agreement (MSA) for litigation involving metal-on-metal hip implants. The settlement resolved 1,292 specifically identified Conserve, Dynasty, or Lineage revision claims. The settlement amounted to $240 million for Wright, but the company estimates that, as of Sept. 25, 2016, there were approximately 600 outstanding metal-on-metal hip revision claims that would not be included in the MSA settlement.
“We are very pleased to have reached this settlement agreement,” commented Palmisano. “This settlement addresses approximately 85 percent of the known U.S. revision claims that do not have potential statute of limitations issues and removes a great deal of the uncertainty that has been associated with this litigation.”
KEY EXECUTIVES:
Robert J. Palmisano, President and CEO
Lance A. Berry, Sr. VP and CFO
Robert P. Burrows, Sr. VP, Supply Chain
J. Wesley Porter, Sr. VP and Chief Compliance Officer
Julie D. Tracy, Sr. VP and Chief Communications Officer
Jennifer S. Walker, Sr. VP, Process Improvement
Kevin D. Cordell, President, U.S.
Peter S. Cooke, President, International
Timothy L. Lanier, President, Upper Extremities
Patrick Fisher, President, Lower Extremities
NO. OF EMPLOYEES: 2,394
GLOBAL HEADQUARTERS: Amsterdam, the Netherlands
Extremities and biologics—two of fastest growing areas of orthopedic technologies. Not coincidentally, these same two areas have become the primary focus for Wright Medical Group. Over the last several years following a series of strategic transactions, the company has positioned itself to focus almost exclusively on these two market sectors.
Beginning in early 2014, Wright Medical divested its hip and knee business (i.e., OrthoRecon) to MicroPort Scientific Corporation. It followed that up with a substantial merger with a leading orthopedic OEM serving the extremities and biologics space—Tornier—which closed Oct. 1, 2015. In 2016 (the fiscal year closed Dec. 25), the company rapidly realized the synergies of that merger, having accomplished more than 80 percent of its approximately 300 integration milestones during the year.
Still seeking to further sharpen its pinpoint focus on the two markets, in July 2016, Wright announced that it was parting ways with its large joints business. Corin Orthopaedics Holdings Ltd. paid approximately $33 million for the hips and knees business. The legacy Tornier unit had become a portion of Wright following the merger. The business sold knee and hip implants primarily in France and other European countries with a product line that included Dynacup and Meije Duo hip implants, as well as HLS KneeTec and HLS Noetos knee implants.
“Our large joints business has excellent products and significant market share in key European markets with a loyal customer base. However, this business is not in line with our strategy to be the premier extremities and biologics company,” Robert Palmisano, president and CEO of Wright Medical, said in a statement regarding the sale. “Post-closing, we will be able to devote our full resources and attention on accelerating growth opportunities in the high-growth extremities and biologics markets and believe this will enhance our ability to create significant shareholder value.”
That focus even permeated the company’s 2016 annual report, with Palmisano kicking it off in his letter with a single-minded message to shareholders, customers, and employees: “Our Focused Excellence approach has enabled us to transform our company to become the leader in the fastest-growing orthopedics markets: upper extremities, lower extremities, and biologics.”
The strategy’s success could be readily observed in the company’s financials for FY16. In the United States (the OEM’s overwhelmingly strongest commercial market), it saw gains in both Upper and Lower Extremities businesses of 14.3 percent and 7.6 percent respectively, over the firm’s 2015 sales in these categories. More impressive was the sales increases seen in the Biologics business. Experiencing a 43.6 percent increase over 2015, the business unit contributed $75 million to Wright’s $690 million total for the year. The company’s fourth business unit—Sports Medicine & Other—remained flat compared to the year prior with $8.4 million in sales. This division offers products used across several anatomic sites to mechanically repair tissue-to-tissue or tissue-to-bone injuries. In many cases, the products complement the upper and lower extremities product portfolios. Overall, the U.S. sales experienced an increase of 14.3 percent over year prior to finish at $507 million.
Primarily responsible for driving the remarkable growth seen in the U.S. Biologics unit was the launch of Wright’s Augment Bone Graft, which was commercially launched in the fourth quarter of 2015. Augment is an alternative to autograft for ankle and/or hindfoot fusion indications, saving the patient and surgeon the time and healing involved with an additional procedure that would be required to harvest the tissue. The biologic product, which was acquired from BioMimetic Therapeutics Inc. in March 2013, had previously faced a rejection from the U.S. Food and Drug Administration (FDA) in 2013 in response to the company’s PMA submission. Two years later, the technology gained the necessary approval and was launched.
“The FDA approval of Augment marks a capstone achievement that demonstrates the strength of our science and provides a breakthrough therapeutic option as an alternative to autograft in ankle and hindfoot fusion procedures,” said Palmisano. “This approval also underscores the significant effort and perseverance from our clinical trial investigators and Wright’s clinical, regulatory, and legal teams to bring the product to market. This success can be attributed to everyone involved in the clinical trial, especially the study investigators and coordinators; our R&D, regulatory, and clinical teams; our partners and suppliers; and most importantly, the patients who participated in the landmark prospective, randomized study comparing Augment Bone Graft to autograft.”
While the company’s international sales aren’t as substantial as those in the United States, they still represent a significant portion of the company’s overall sales for the year. Growth, unfortunately, was not as optimistic as the U.S. sales either, but overall, international still saw a 6.6 percent gain over 2015 to total $183 million. That number breaks down in a similar manner to the U.S. splits, with Lower Extremities ($63 million) and Upper Extremities ($87 million) representing the bulk of the company’s business. Those sales totals represent a 6.7 percent increase for Lower Extremities over 2015, while Upper Extremities experienced the greatest growth among international sales between the business units at 11.8 percent compared to FY15. Biologics ($19 million) and Sports Medicine & Other ($15 million), on the other hand, both saw sales decline in international markets by 5.7 percent and 4.1 percent respectively, compared to the prior year.
Before wrapping up what had amounted to a relatively successful year in terms of market growth and sales increases, new product launches, and gained efficiencies from the 2015 Tornier merger, Wright Medical resolved some pending legal issues with regard to its wholly owned subsidiary Wright Medical Technology.
The company entered into a Master Settlement Agreement (MSA) for litigation involving metal-on-metal hip implants. The settlement resolved 1,292 specifically identified Conserve, Dynasty, or Lineage revision claims. The settlement amounted to $240 million for Wright, but the company estimates that, as of Sept. 25, 2016, there were approximately 600 outstanding metal-on-metal hip revision claims that would not be included in the MSA settlement.
“We are very pleased to have reached this settlement agreement,” commented Palmisano. “This settlement addresses approximately 85 percent of the known U.S. revision claims that do not have potential statute of limitations issues and removes a great deal of the uncertainty that has been associated with this litigation.”