Bernd Robioneck, Stryker Orthopaedics 08.31.10
The collaboration between experienced surgeons and the engineers of device manufacturers has been the foundation of our industry and has resulted in product innovations that have moved the whole industry forward. Today, implants last longer and provide the opportunity for better surgical outcomes, and new instrumentation helps to boost efficiency in the operating room.
One of the primary challenges that medical device makers always have faced was how to address the broadest patient population possible with standardized orthopedic products and instruments. Every patient’s anatomy is unique, and, as a result, orthopedic products commonly are not “one size fits all.”
Historically, surgeons who worked with medical device makers’ engineers to develop implants designed them based on the anatomy of a limited number of cadaver specimens. While this enabled products to be developed based on real patient anatomy, the cadavers upon whom the devices were designed often were not a large sample group that was truly representative of the full range of patient anatomies.
During the implant selection process, surgeons commonly have been required to spend time in the operating room (OR) sizing a patient’s anatomy, and then selecting the implant with the most appropriate size to ensure the most optimal fit. Not only did this process reduce operating room efficiency, it also meant that surgeons needed to stock a wide inventory of implants, cutting down on valuable OR space.
Armed with decades of clinical data and advanced computer programming, orthopedic device makers today have uncovered a way to manufacture implants that fit a greater range of patient anatomies, helping to boost OR efficiency. Surgeons and device makers now can design an implant using computer databases filled with the CT scans of patients’ joints, which enables them to use “evidence-based design” to create implants capable of addressing the greatest possible patient population.1
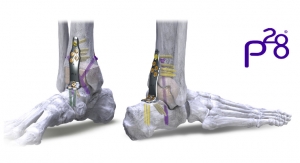
Trauma surgery is one great example of the value that evidence-based design brings to surgeons and hospitals. Trauma surgeons have always needed to have two sets of orthopedic plates to repair patients’ feet and ankles, one for the left side of the body and one for the right. These plates commonly were straight, and, as a result, surgeons needed to spend a significant period of time in the OR shaping the plate in order to fit an individual patient’s anatomy.
By using its virtual database of CT scans, Stryker was able to create VariAx trauma plating implants, which universally are designed to fit the bones on patients’ right and left sides of the body. In addition, the plating implants are pre-contoured so that they match the large majority of patient anatomies. This change in design boosts OR efficiency and reduces costs by enabling surgeons to spend less time shaping the implants, with the goal of having patients spend less time lying on the OR table. It also reduces the amount of inventory that hospitals and surgery practices need to store.
This design of plating products also enables surgeons potentially to enhance the clinical outcomes of patients with fibular fractures. Stryker’s VariAx Fibula plates have a “spoon” shaped end. Using a straight plate limits a surgeon’s availability to repair a patient’s distal fibula, but with the unique anatomical design of devices such as the VariAx Fibula plate, surgeons can capture and reduce bone fragments, effectively treating difficult fractures.
The plates are manufactured using high-quality titanium, which enables the design to incorporate poly-axial locking. Poly-axial locking provides surgeons with the ability to choose the best angles at which screws can be angulated within a plate, and by extension, into bone. Furthermore, poly-axial locking relies on the plate-screw interface for construct strength and stability, rather than a bone-screw interface, which was used in implants of the past.
These kinds of advances are only a few examples of the value that virtual databases and evidence-based design can bring to the healthcare industry as a whole. As the orthopedic industry moves forward, device makers need to continue to capitalize on the use of virtual databases to inform the device development process.
The collaboration between surgeons and engineers will continue to drive implant innovation in the orthopedics industry. But it is the use of new development capabilities, such as evidence-based design, that will help to ensure this collaboration results in the most effective products that bring maximum efficiency to stakeholders throughout healthcare.
Reference:
1. Nina Kozic, Stefan Weber, et al. (2010) Optimisation of orthopaedic implant design using statistical share space analysis based on level sets. Medical Image Analysis. 265-275.
Bernd Robioneck is vice president of Research and Development and Technical Marketing for Stryker Orthopaedics.
One of the primary challenges that medical device makers always have faced was how to address the broadest patient population possible with standardized orthopedic products and instruments. Every patient’s anatomy is unique, and, as a result, orthopedic products commonly are not “one size fits all.”
Historically, surgeons who worked with medical device makers’ engineers to develop implants designed them based on the anatomy of a limited number of cadaver specimens. While this enabled products to be developed based on real patient anatomy, the cadavers upon whom the devices were designed often were not a large sample group that was truly representative of the full range of patient anatomies.
During the implant selection process, surgeons commonly have been required to spend time in the operating room (OR) sizing a patient’s anatomy, and then selecting the implant with the most appropriate size to ensure the most optimal fit. Not only did this process reduce operating room efficiency, it also meant that surgeons needed to stock a wide inventory of implants, cutting down on valuable OR space.
Armed with decades of clinical data and advanced computer programming, orthopedic device makers today have uncovered a way to manufacture implants that fit a greater range of patient anatomies, helping to boost OR efficiency. Surgeons and device makers now can design an implant using computer databases filled with the CT scans of patients’ joints, which enables them to use “evidence-based design” to create implants capable of addressing the greatest possible patient population.1
Trauma surgery is one great example of the value that evidence-based design brings to surgeons and hospitals. Trauma surgeons have always needed to have two sets of orthopedic plates to repair patients’ feet and ankles, one for the left side of the body and one for the right. These plates commonly were straight, and, as a result, surgeons needed to spend a significant period of time in the OR shaping the plate in order to fit an individual patient’s anatomy.
By using its virtual database of CT scans, Stryker was able to create VariAx trauma plating implants, which universally are designed to fit the bones on patients’ right and left sides of the body. In addition, the plating implants are pre-contoured so that they match the large majority of patient anatomies. This change in design boosts OR efficiency and reduces costs by enabling surgeons to spend less time shaping the implants, with the goal of having patients spend less time lying on the OR table. It also reduces the amount of inventory that hospitals and surgery practices need to store.
This design of plating products also enables surgeons potentially to enhance the clinical outcomes of patients with fibular fractures. Stryker’s VariAx Fibula plates have a “spoon” shaped end. Using a straight plate limits a surgeon’s availability to repair a patient’s distal fibula, but with the unique anatomical design of devices such as the VariAx Fibula plate, surgeons can capture and reduce bone fragments, effectively treating difficult fractures.
The plates are manufactured using high-quality titanium, which enables the design to incorporate poly-axial locking. Poly-axial locking provides surgeons with the ability to choose the best angles at which screws can be angulated within a plate, and by extension, into bone. Furthermore, poly-axial locking relies on the plate-screw interface for construct strength and stability, rather than a bone-screw interface, which was used in implants of the past.
These kinds of advances are only a few examples of the value that virtual databases and evidence-based design can bring to the healthcare industry as a whole. As the orthopedic industry moves forward, device makers need to continue to capitalize on the use of virtual databases to inform the device development process.
The collaboration between surgeons and engineers will continue to drive implant innovation in the orthopedics industry. But it is the use of new development capabilities, such as evidence-based design, that will help to ensure this collaboration results in the most effective products that bring maximum efficiency to stakeholders throughout healthcare.
Reference:
1. Nina Kozic, Stefan Weber, et al. (2010) Optimisation of orthopaedic implant design using statistical share space analysis based on level sets. Medical Image Analysis. 265-275.
Bernd Robioneck is vice president of Research and Development and Technical Marketing for Stryker Orthopaedics.