07.31.12
$540 Million
KEY EXECUTIVES:
Alexis V. Lukianov, Chairman and CEO
Keith C. Valentine, President and Chief Operating Officer
Patrick Miles, President, Global Products and Services
Tyler P. Lipschultz, Exec. VP, Global Operations
G. Bryan Cornwall, Ph.D., Sr. VP, Research and Clinical Resources
Russell Powers, Sr. VP, Operations
NO. OF EMPLOYEES: 1,500
GLOBAL HEADQUARTERS: San Diego, Calif.
If you ask NuVasive Inc. employees for the secret to the company’s success, they’re likely to repeat the firm’s mantra it takes the combination of “speed of innovation; absolute responsiveness; and superior clinical outcomes.” That’s pretty much a winning formula in any medical device manufacturer’s book, but it’s not easy to get all those proverbial parts moving in the same direction. At least a little something must be working well with a 13 percent increase in revenue for fiscal 2011 (ended Dec. 31, 2011).
It’s no secret that the Southern California-based spinal firm aggressively has been chasing market share. By the company’s estimates, it is the fifth-largest player in the nearly $8 billion global spine market.
“We have our sights set on becoming a $1 billion ‘start up,’ where we can offer the speed and passion of a start up, with the experience and resources of a larger company,” CEO Alex Lukianov said.
They’re a little more than halfway there. Sales for the fiscal year were just shy of $541 million (which put it—for the first time—on Orthopedic Design & Technology’s Top 10 list).
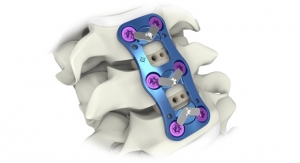
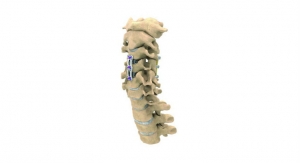
The company’s principal product offering is based on its Maximum Access Surgery (MAS) platform, which is designed to minimize soft tissue disruption during spine surgery with improved visualization and safe, and reproducibility for the surgeon; a proprietary software-driven nerve avoidance system and intra-operative monitoring support; MaXcess, a unique split-blade retractor system; a variety of specialized implants; and several biologic fusion enhancers. With the MAS platform’s lateral approach, which is dubbed eXtreme Lateral Interbody Fusion (XLIF), NuVasive has built an entire spine franchise. At present, the company has more than 70 products spanning lumbar, thoracic and cervical applications.
Gross profit for the full year 2011 was $428 million and gross margin was 79.3 percent, compared to a gross profit of $393 million and gross margin of 82.2 percent for the full year 2010. Full year 2011 operating expenses were $513 million compared to $361 million reported for 2010. The higher operating expenses in 2011 resulted primarily from a charge related to intellectual property litigation, an intangible asset impairment charge, and additional costs associated with higher revenue and infrastructure expansion. On the basis of generally accepted accounting principles, the company reported a net loss of nearly $70 million, or $1.73 per share for the full year 2011. Cash, cash equivalents and short and long-term marketable securities were $342 million at the end of the year.
Many of the current market challenges in the U.S. spine market are the result of several commercial insurance providers increasing the level of scrutiny and the enforcement of criteria necessary for spine patients to receive reimbursement preauthorization for lumbar fusion procedures.
To further expand its reach—in addition to a robust research and development budget—NuVasive acquired Columbia, Md.-based Impulse Monitoring Inc. in October, which provides the firm with a comprehensive intra-operative monitoring (IOM) solution. NuVasive paid $80 million, ($40 million in cash and $40 million in stock).
“The integration of Impulse will open up an incremental per procedure revenue opportunity in a market that is growing about 15 percent annually, and while that aspect alone is encouraging, the strategic rationale behind the combination will play out over the long term,” said Lukianov at the time the deal was announced. “Our team of neurophysiologists, who are exceptionally valued members of their operating room teams, can help surgeons fundamentally appreciate the power of integrated neuromonitoring and our highly differentiated and broad product portfolio. Combined with our focus on superior clinical outcomes, we believe this strategy will drive surgeon conversion and accelerate our ability to take market share.”
Intra-operative monitoring technology allows procedural detection of neurological problems and identification of functional neural structures during surgeries that involve spine; cardio; ear, nose and throat; brain; and general orthopedic. The IOM model employs highly trained neurophysiologists located in the operating room who function as part of the surgical team, assisting with patient preparation, administering neuro-diagnostic testing, and working in partnership with supervising physicians who provide real time interpretation of the neurophysiological data. Neuromonitoring of the spine was 80 percent of Impulse Monitoring’s case volume.
This acquisition will add to NuVasive’s NeuroVision nerve monitoring system, which is designed for discreet and directional nerve avoidance and detection, making lateral access to the spine during the XLIF procedure safer and more reproducible.
According to NuVasive there is $800 million market for intra-operative monitoring. Today, only 50 percent of spine procedures undergo IOM. Over the next several years, the market for outsourced IOM is expected to grow approximately 15 percent annually, the firm predicts.
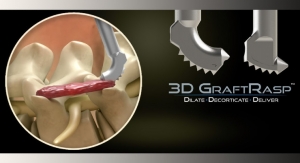
In 2011, the company launched the next generation of its MaXcess retractor (a split-blade design retraction system providing what the company claims is improved surgical access to the spine) with integrated neuromonitoring; a software upgrade for NeuroVision nerve avoidance system; and a series of new indication-specific implants, including the X-Core expandable cage for corpectomies (a surgical procedure that involves removing part of the vertebral body—or corpus vertebrae in Latin, hence the name corpectomy—usually as a way to decompress the spinal cord and nerves).
You usually can’t have increased sales without a strong sales force. NuVasive’s increased its sales force in 2011, adding more sales representatives both domestically and internationally. With a footprint in more than 35 countries, 2011 marked the first year that the company’s international operations broke even from a profitability perspective. NuVasive is now in the second-largest spine market outside of the United States with operations in Japan, which began in this year. In addition to corporate headquarters in San Diego, the company has additional offices in Memphis, Tenn.; Paramus, N.J.; Puerto Rico; the United Kingdom; Germany; Australia; Singapore; and Malaysia.
During the year NuVasive licensed to Centennial, Colo.-based AlloSource certain intellectual property related to bone growth products containing endogenous mesenchymal stem cells. The license and royalty agreement allows AlloSource to distribute its AlloStem allograft cellular product. The agreement includes restrictions against AlloSource distributing into the spine market with a major partner other than NuVasive. AlloSource currently processes Osteocel Plus for NuVasive. AlloSource is a non-profit company that offers more than 200 precise bone, skin, soft-tissue and custom-machined allografts for use in an array of life-saving and life-enhancing medical procedures. It provides cartilage tissue used for joint repair and skin allografts to heal severe burns, and is one of the largest tissue networks, as well as the largest processor of live cellular bone growth substitutes.
Not all the news was rosy during the year, however. Medtronic won a $101.2 million jury award in a patent-infringement trial against NuVasive. The federal jury also held Medtronic liable for $660,000 in damages to NuVasive for infringing one of its patents. In a two-week trial, Warsaw Orthopedic Inc., a unit of Minneapolis-based Medtronic, accused NuVasive of infringing three patents for implants capable of being inserted trans-laterally between adjacent vertebrae, a plate and screw system used to stabilize vertebrae in the cervical spine and a tissue retractor, according to court filings. Though NuVasive leadership insisted they hadn’t copied anything and vowed to challenge the verdict, NuVasive’s requests to overturn the Sept. 20 jury verdict or order a new trial was denied by the court. The damages awarded to Medtronic were the 18th-largest jury award in the United States in 2011 and the fourth-largest in a patent-infringement claim, according to data compiled by Bloomberg. The judge, however, denied Medtronic’s request for a permanent injunction barring NuVasive from selling the devices found to infringe its patents. He also left intact the jury’s award of $660,000 in damages to NuVasive for Medtronic’s infringement of one of its patents.
In 2012, NuVasive—along with most of the medical device industry—has been active and vocal in its opposition of the 2.3 percent excise tax slated to take effect in 2013 as part of the healthcare reform legislation passed in 2010 and recently upheld by the U.S. Supreme Court. Lukianov recently wrote an op-ed for the San Diego Union Tribune newspaper highlighting the damage the medical device tax and universal healthcare reportedly would do to his company. Lukianov wrote that 100 percent of R&D is currently done within the United States, as well as 90 percent of all manufacturing. He added that he might need to layoff 200 of his 1,500 employees in 2013, and potentially move parts of his R&D and manufacturing process to more tax-favorable countries.
KEY EXECUTIVES:
Alexis V. Lukianov, Chairman and CEO
Keith C. Valentine, President and Chief Operating Officer
Patrick Miles, President, Global Products and Services
Tyler P. Lipschultz, Exec. VP, Global Operations
G. Bryan Cornwall, Ph.D., Sr. VP, Research and Clinical Resources
Russell Powers, Sr. VP, Operations
NO. OF EMPLOYEES: 1,500
GLOBAL HEADQUARTERS: San Diego, Calif.
If you ask NuVasive Inc. employees for the secret to the company’s success, they’re likely to repeat the firm’s mantra it takes the combination of “speed of innovation; absolute responsiveness; and superior clinical outcomes.” That’s pretty much a winning formula in any medical device manufacturer’s book, but it’s not easy to get all those proverbial parts moving in the same direction. At least a little something must be working well with a 13 percent increase in revenue for fiscal 2011 (ended Dec. 31, 2011).
It’s no secret that the Southern California-based spinal firm aggressively has been chasing market share. By the company’s estimates, it is the fifth-largest player in the nearly $8 billion global spine market.
“We have our sights set on becoming a $1 billion ‘start up,’ where we can offer the speed and passion of a start up, with the experience and resources of a larger company,” CEO Alex Lukianov said.
They’re a little more than halfway there. Sales for the fiscal year were just shy of $541 million (which put it—for the first time—on Orthopedic Design & Technology’s Top 10 list).
The company’s principal product offering is based on its Maximum Access Surgery (MAS) platform, which is designed to minimize soft tissue disruption during spine surgery with improved visualization and safe, and reproducibility for the surgeon; a proprietary software-driven nerve avoidance system and intra-operative monitoring support; MaXcess, a unique split-blade retractor system; a variety of specialized implants; and several biologic fusion enhancers. With the MAS platform’s lateral approach, which is dubbed eXtreme Lateral Interbody Fusion (XLIF), NuVasive has built an entire spine franchise. At present, the company has more than 70 products spanning lumbar, thoracic and cervical applications.
Gross profit for the full year 2011 was $428 million and gross margin was 79.3 percent, compared to a gross profit of $393 million and gross margin of 82.2 percent for the full year 2010. Full year 2011 operating expenses were $513 million compared to $361 million reported for 2010. The higher operating expenses in 2011 resulted primarily from a charge related to intellectual property litigation, an intangible asset impairment charge, and additional costs associated with higher revenue and infrastructure expansion. On the basis of generally accepted accounting principles, the company reported a net loss of nearly $70 million, or $1.73 per share for the full year 2011. Cash, cash equivalents and short and long-term marketable securities were $342 million at the end of the year.
Many of the current market challenges in the U.S. spine market are the result of several commercial insurance providers increasing the level of scrutiny and the enforcement of criteria necessary for spine patients to receive reimbursement preauthorization for lumbar fusion procedures.
To further expand its reach—in addition to a robust research and development budget—NuVasive acquired Columbia, Md.-based Impulse Monitoring Inc. in October, which provides the firm with a comprehensive intra-operative monitoring (IOM) solution. NuVasive paid $80 million, ($40 million in cash and $40 million in stock).
“The integration of Impulse will open up an incremental per procedure revenue opportunity in a market that is growing about 15 percent annually, and while that aspect alone is encouraging, the strategic rationale behind the combination will play out over the long term,” said Lukianov at the time the deal was announced. “Our team of neurophysiologists, who are exceptionally valued members of their operating room teams, can help surgeons fundamentally appreciate the power of integrated neuromonitoring and our highly differentiated and broad product portfolio. Combined with our focus on superior clinical outcomes, we believe this strategy will drive surgeon conversion and accelerate our ability to take market share.”
Intra-operative monitoring technology allows procedural detection of neurological problems and identification of functional neural structures during surgeries that involve spine; cardio; ear, nose and throat; brain; and general orthopedic. The IOM model employs highly trained neurophysiologists located in the operating room who function as part of the surgical team, assisting with patient preparation, administering neuro-diagnostic testing, and working in partnership with supervising physicians who provide real time interpretation of the neurophysiological data. Neuromonitoring of the spine was 80 percent of Impulse Monitoring’s case volume.
This acquisition will add to NuVasive’s NeuroVision nerve monitoring system, which is designed for discreet and directional nerve avoidance and detection, making lateral access to the spine during the XLIF procedure safer and more reproducible.
According to NuVasive there is $800 million market for intra-operative monitoring. Today, only 50 percent of spine procedures undergo IOM. Over the next several years, the market for outsourced IOM is expected to grow approximately 15 percent annually, the firm predicts.
In 2011, the company launched the next generation of its MaXcess retractor (a split-blade design retraction system providing what the company claims is improved surgical access to the spine) with integrated neuromonitoring; a software upgrade for NeuroVision nerve avoidance system; and a series of new indication-specific implants, including the X-Core expandable cage for corpectomies (a surgical procedure that involves removing part of the vertebral body—or corpus vertebrae in Latin, hence the name corpectomy—usually as a way to decompress the spinal cord and nerves).
You usually can’t have increased sales without a strong sales force. NuVasive’s increased its sales force in 2011, adding more sales representatives both domestically and internationally. With a footprint in more than 35 countries, 2011 marked the first year that the company’s international operations broke even from a profitability perspective. NuVasive is now in the second-largest spine market outside of the United States with operations in Japan, which began in this year. In addition to corporate headquarters in San Diego, the company has additional offices in Memphis, Tenn.; Paramus, N.J.; Puerto Rico; the United Kingdom; Germany; Australia; Singapore; and Malaysia.
During the year NuVasive licensed to Centennial, Colo.-based AlloSource certain intellectual property related to bone growth products containing endogenous mesenchymal stem cells. The license and royalty agreement allows AlloSource to distribute its AlloStem allograft cellular product. The agreement includes restrictions against AlloSource distributing into the spine market with a major partner other than NuVasive. AlloSource currently processes Osteocel Plus for NuVasive. AlloSource is a non-profit company that offers more than 200 precise bone, skin, soft-tissue and custom-machined allografts for use in an array of life-saving and life-enhancing medical procedures. It provides cartilage tissue used for joint repair and skin allografts to heal severe burns, and is one of the largest tissue networks, as well as the largest processor of live cellular bone growth substitutes.
Not all the news was rosy during the year, however. Medtronic won a $101.2 million jury award in a patent-infringement trial against NuVasive. The federal jury also held Medtronic liable for $660,000 in damages to NuVasive for infringing one of its patents. In a two-week trial, Warsaw Orthopedic Inc., a unit of Minneapolis-based Medtronic, accused NuVasive of infringing three patents for implants capable of being inserted trans-laterally between adjacent vertebrae, a plate and screw system used to stabilize vertebrae in the cervical spine and a tissue retractor, according to court filings. Though NuVasive leadership insisted they hadn’t copied anything and vowed to challenge the verdict, NuVasive’s requests to overturn the Sept. 20 jury verdict or order a new trial was denied by the court. The damages awarded to Medtronic were the 18th-largest jury award in the United States in 2011 and the fourth-largest in a patent-infringement claim, according to data compiled by Bloomberg. The judge, however, denied Medtronic’s request for a permanent injunction barring NuVasive from selling the devices found to infringe its patents. He also left intact the jury’s award of $660,000 in damages to NuVasive for Medtronic’s infringement of one of its patents.
In 2012, NuVasive—along with most of the medical device industry—has been active and vocal in its opposition of the 2.3 percent excise tax slated to take effect in 2013 as part of the healthcare reform legislation passed in 2010 and recently upheld by the U.S. Supreme Court. Lukianov recently wrote an op-ed for the San Diego Union Tribune newspaper highlighting the damage the medical device tax and universal healthcare reportedly would do to his company. Lukianov wrote that 100 percent of R&D is currently done within the United States, as well as 90 percent of all manufacturing. He added that he might need to layoff 200 of his 1,500 employees in 2013, and potentially move parts of his R&D and manufacturing process to more tax-favorable countries.