Ranica Arrowsmith, Associate Editor08.05.13
While giants such as Johnson & Johnson and Stryker Corp. continue to dominate the orthopedic device market, smaller, younger companies are capitalizing on the sub-segments of orthopedics that are most promising. The orthopedics market is growing as baby boomers in America age and populations in BRIC countries, particularly China, become more affluent. Niche markets such as regenerative medicine are ready and waiting for the right companies to occupy space within them.
According to ASG Reports, a market research and analysis firm based in Amsterdam, The Netherlands, the global orthopedics devices market is forecast to reach a value of $61.3 billion by 2017 from a market value of $36.7 billion in 2010 at a compound annual growth rate of 7.6 percent. The report identified minimally invasive devices as being the driver behind the new technologies market. Companies such as Baxano Surgical (formerly Trans1) wisely are anticipating this trend by creating a strong foothold and a robust portfolio in the minimally invasive surgery (MIS) orthopedics market.
Finding—and creating—niche markets is becoming essential to survival for smaller medical device companies. While the large medical device companies such as DePuy Synthes, Stryker, Zimmer Holdings Inc., Smith & Nephew plc and Medtronic Inc. together hold more than 60 percent of market share in the global orthopedic devices market, smaller companies can maintain a strong foothold if they innovate and stick to what they know best. EOS Imaging, for instance, has harnessed Nobel Prize-winning technology to create a unique orthopedic imaging system that no other company really has a finger in.
“Decreasing profit margins and pressure to launch new products has led to consolidation,” read an ASG report on orthopedic market growth published in March last year. “For example, in 2011 DePuy acquired Synthes. Furthermore, increasing competition from local companies with low cost products has led to an increased focus on biomaterials and orthobiologics, meaning that these products could hold the key to growth in the orthopedic devices industry.”
Orthobiologics, which includes regenerative medicine, deals with growing new human tissue instead of using artificial implants to heal fractures and other orthopedic ailments. Companies such as ISTO Technologies and Bioventus are harnessing stem cell technology and ultrasound pulses to promote and accelerate the body healing itself.
EOS Imaging
Location: Paris, France
Leadership: Marie Meynadier, Ph.D., CEO
Sector: Orthopedic imaging
Online: www.eos-imaging.com
EOS Imaging SA is built upon a technology that won the 1992 Nobel Prize for Physics: a high energy particle detector. Georges Charpak’s invention detected “radiations that help understand the origin of the universe,” explained ESO CEO Marie Meynadier, Ph.D., to Orthopedic Design & Technology; and EOS has harnessed that capability in its unique imaging system that can take three dimensional images of human bone in weight-bearing positions, all with a very low radiation dose.
Meynadier, who has a background in microelectronics, started EOS in 2005 as a young startup by the name of Biospace Med. The company changed its name to EOS Imaging in 2010 when the EOS brand really took off and gained recognition. “Doctors started asking for ‘an EOS exam,’ not a ‘pair of X-rays,’” said Meynadier. By 2010, the EOS imaging system had become as recognizable to those using it as a CT (computed tomography) or MRI (magnetic resonance imaging) scan.
The EOS System, which EOS manufactures, provides exactly that—a set of very low dose X-rays from which 3-D imaging of the skeleton is derived. When Meynadier met Charpak, he had a groundbreaking technology that he needed to market, and Meynadier had an idea of where his particle detector could be best used. Bone imaging presents unique challenges: Physicians and medical professionals often need to view large sections of the patient’s body at once, such as whole legs or whole spines, and the radiation associated with these exams was high. Besides, traditional X-rays only give a single-angle, “flat” view. CT and MRI machines can provide 3-D imaging, but when the patient’s body is recumbent, the joints are not being viewed in a functional position. Also, CT scans place the body under high doses of radiation, while the EOS system is emits very low doses of the harmful rays.
“EOS takes two radiographs of the patient, frontal and lateral simultaneously,” explained Meynadier. “It does it at a very low dose and it has the ability to do that over the whole body of a patient. Then, with software, we can reconstruct an envelope of the patient’s bones in 3-D in a weight bearing position, and extract a full set of clinical parameters that the surgeons want, whether it’s in spine, hip or knee surgery.”
EOS’ growth has exploded in the past two years. Its revenues in 2012 were $9.14 million, 36 percent above the year before. Already for the first quarter of 2013, revenues are $2.49 million, while for the same quarter last year they were $730,000. Sixty-two percent of revenues in Q1 2013 were generated from North America, where the company sold three EOS systems. In Europe, revenues were $790,000, which included the sale of one EOS system.
2012 was a landmark year for EOS Imaging. The company gained significant financial resources from an initial public offering in February, which allowed it to make “controlled commercial investments to bolster growth in 2013 and beyond,” according to Meynadier. Last year, the company also saw EOS adopted in four of the top seven U.S. orthopedic hospitals. The United States will be a key focus region for future growth, and EOS has offices in both Cambridge, Mass., and Montreal, Canada. EOS also
recently opened an office in Singapore to serve the Asian market.
EOS manufacturing operations are located in France, and the company has no plans to move manufacturing anywhere else. “There are lots of non-quality issues linked to manufacturing everywhere on the planet,” Meynadier said. “At our size, for the years to come, we believe that our manufacturing has been strongly secured; keeping manufacturing close to our headquarters is the best option for EOS.”
EOS has no plans to work on different products in the future; rather, the company will continue to focus on expanding the capabilities of the EOS system.
“There are a large number of avenues of usage in the orthopedic practice that we are investigating with surgeon partners,” Meynadier said. “Not only with diagnostics, but also planning, simulation and quality control. We are more focused on developing more software features for EOS than developing new devices. Quality control for hip and knee surgery, for instance, is extremely straightforward with the EOS system. This is a very strong asset to an accountable care organization for its ability to plan a given surgery, execute it and control its quality with a reliable tool.”
Baxano Surgical
Location: Raleigh, N.C.
Leadership: Ken Reali, President and CEO
Sector: Degenerative conditions of lumbar spine
Online: www.baxanosurgical.com
At the end of May this year, Trans1 Inc. completed the acquisition of Baxano Inc. and became
Baxano Surgical Inc. The newly branded company describes itself as a medical device company focused on products to treat degenerative conditions of the spine affecting the lumbar region. Baxano Surgical currently markets the AxiaLIF family of products for single and two level lumbar fusion, the VEO lateral access and interbody fusion system, and the iO-Flex system, a proprietary minimally invasive set of flexible instruments allowing surgeons to target lumbar spinal stenosis during spinal decompression procedures in all three regions of the spine: central canal, lateral recess, and neural foramen.
The merger was part of a four-part plan that was conceived in 2010 when Ken Reali joined Trans1 as CEO. First, the company needed to cut its burn rate and work towards establishing positive cash flow. To help this goal, Trans1 needed to establish physician reimbursement for AxiaLIF, which it did Jan. 1 this year. Thirdly, the company launched Veo early last year. The final part of the plan was an acquisition.
“We’ve had our eye on Baxano since 2010,” Reali told ODT. “We really liked the iO-Flex technology and we saw a direct fit and synergy with our fusion systems AxiaLIF and Veo. The acquisition not only gives us a timeline to get to profitability, but shortens our timeline to profitability by a year.”
The company was founded as Trans1 in 2000, when Andy Cragg, M.D., an interventional radiologist in Minneapolis, Minn., had the idea for a pre-sacral approach to the lumbar spine. Cragg’s brother, an orthopedic surgeon, observed that the damage he would do to the soft tissue structures through traditional access to the lumbar spine seemed to cause more pain to the patient than their original back pain. Recovery periods were long and this gave lumbar fusion surgery a bad reputation. Cragg’s own experience in interventional radiology, which is concerned with using minimally invasive image-guided procedures to diagnose and treat diseases in nearly every organ system, influenced him to consider the spinal column as a tubular structure. With this vision, he developed a method of accessing the lumbar spine via the spine’s axis rather than perpendicularly from the side, front, or back. Cragg then solicited the help of engineer Bob Assell who took the physician’s catheter thinking and turned it into proprietary instrumentation that accomplished the task while retaining a design familiar to orthopedic and neuro surgeons.
This instrumentation eventually became Baxano’s AxiaLIF system: Nitinol was used in the construction of special cutters designed to debulk the nucleus of the disc and rough up the vertebral endplates; and stainless steel tissue extractors were created to remove the disc material so that bone graft could be inserted into the disc space. In 2004, AxiaLIF earned 510(k) clearance from the U.S. Food and Drug Administration (FDA). The very next year, Baxano Inc. was founded. Baxano was founded by Jeff Bleich, M.D., a physician at Stanford University Medical Center, who had recognized the significant clinical need for a less invasive, less traumatic means of achieving a lumbar spinal decompression.
Together with serialentrepreneur Vahid Saadat, Bleich led a team of clinical advisors and engineers to develop a set of modified rongeurs (instruments with a sharp-edged, scoop-shaped tip, used for gouging out bone) that would become the first iO-Flex prototypes.
In 2007, Baxano received FDA clearance to market the iO-Flex Microblade Shaver. The company gained a U.S. patent on the system in 2009, and named Tony Recupero CEO for his extensive experience in sales the same year. Baxano was focused on aggressively expanding its intellectual property portfolio, and Recupero was the man for the job. Last year, iO-Flex earned CE mark and Health Canada approval to market in European Union countries and Canada.
“The synergies of multiple lines of differentiated MIS technologies, serving specialists within spine surgery focusing on MIS makes strategic sense to us, and the deal also scales up the growth model significantly for the company over the next two to three years,” wrote Piper Jaffray analyst Matt Miksic upon news of the merger. “In addition to its strategic positives, the financing associated with the deal will delay (but not eliminate) the need to raise additional cash, easing an overhang on TranS1 since January.”
The future of Baxano Surgical is all about new products. The company’s new minimally invasive facetectomy removal instrument, iO-Tome, was cleared by the FDA in October last year, and Baxano plans to release it in the fourth quarter of 2013. The instrument is designed to rapidly and precisely remove the facet joint, a procedure commonly required in spinal fusion applications. In the pipeline for next year is a minimally invasive pedicle screw system called Avance.
“We will exit this year with four flagship products, all proprietary and differentiated, and all focused in the minimally invasive spine arena,” said Reali.
Bioventus
Location: Durham N.C.
Leadership: Mark Augusti, CEO
Sector: Orthobiologics
Online: www.bioventusglobal.com
Bioventus LLC is a very young company, having been founded just last year. However, the company spun off from the Biologics and Clinical Therapies division of one of the largest orthopedic companies in the world, Smith & Nephew, so it was born with a considerable advantage. The spinoff was a strategic venture between an Essex Woodlands-led investor syndicate group and Smith & Nephew.
The company’s flagship product is Exogen, a system that uses ultrasound signals to accelerate bone healing. It is the only U.S. Food and Drug Administration-approved device that uses a low intensity pulsed ultrasound signal for bone healing. It is indicated for use in non-union fractures (fractures that have not healed properly) and certain kinds of stress fractures.
Bioventus also markets Supartz in the United States. This is a three- to five-injection regimen for the treatment of mild to moderate osteoarthritis pain in patients who do not respond to steroids or analgesics. Outside the United States, the company markets Durolane, which is a single injection technology for the same indication. The company intends to bring Durolane stateside, and is working with U.S. regulatory authorities to begin clinical trials and the process for approval of the product.
The products Bioventus makes fall into the category of, as CEO Mark Augusti describes it, “active orthopedic healing.” This kind of technology, Augusti explained last year to ODT, is often described as orthobiologics. Studies show, however, that this type of healing has a sociological effect, which is why Bioventus brands its approach a little differently.
Upon its founding, Bioventus was still operating under the name Smith & Nephew outside the United States. Smith & Nephew was working essentially as the company’s distributor; changing the branding would happen gradually as Bioventus navigated various countries’ labor regulations. In December last year, Bioventus opened its international headquarters in The Netherlands just outside Amsterdam, which enabled Bioventus’ products distributed there to be transitioned to the Bioventus brand. Italy was next to receive the Bioventus brand and products; then the United Kingdom, Ireland, Sweden, Norway, Denmark, Canada, France, Finland, and most recently, Germany.
When Augusti took the reigns as CEO of Bioventus at its inception, he was already a seven-year veteran of Smith & Nephew, and was serving as president of the company’s Biologics & Spine business. Key appointments since then have been David Price as chief financial officer, who took on the role in October last year; and Peter Heeckt, M.D., Ph.D., as chief medical officer, appointed in April this year. Price joined Bioventus from EDGAR Online Inc., a financial data, technology and business process outsourcing company; and Heeckt most recently was co-founder and managing member for Life Sciences Consulting Services LLC, a consulting firm specializing in medical and clinical affairs and healthcare compliance for medical device and biotech companies.
Bioventus is earning more global street cred as its products become recognized by international regulatory and other bodies. In May this year, the NHS (National Health Service) Technology Adoption Centre in the United Kingdom, the implementation arm of the National Institute for Health and Care Excellence (NICE), recently launched a new adoption pack regarding Exogen. The adoption pack is designed to help NHS trusts correctly implement guidelines NICE released supporting the use of Exogen as an alternative to surgery for thousands of patients with long bone non-union fractures. The pack includes information about the benefits of Exogen to patients, physicians and healthcare systems, the national and local opportunities available, the drivers for adoption, detailed case studies of where Exogen has already been successfully used in the NHS, and the costs and potential savings.
ISTO Technologies
Location: St. Louis, Mo.
Leadership: Mitchell Seyedin, Ph.D., President and CEO
Sector: Orthobiologics
Online: www.istotech.com
Regenerative medicine is an exciting new field in orthopedics, not least because of its challenges. Vague laws and very young research make the field of tissue regeneration difficult to navigate, but dynamic St. Louis, Mo.-based orthobiologics company ISTO Technologies Inc. has been working since 2002 to perfect cartilage regeneration, which is particularly problematic. Finding stem cells that can regenerate cartilage reliably without fusing with and turning into hard bone down the line has proved very difficult. Recent research has shown that children younger than one year of age have much more of a cell population capable of regenerating cartilage than anyone else. ISTO holds patents on juvenile cell-based technology that they use in their products.
ISTO began operations in 2002 by sharing the development responsibilities of Denovo ET (engineered tissue) with Zimmer Holdings. This year, just over a decade later, ISTO took full control of the technology. Denovo ET is an engineered cartilage implant designed to repair defects in the knee.
ISTO renamed the product Revaflexand currently is proceeding independently with further development of the program through a Phase III clinical program. The technology harnesses juvenile cartilage cells to create a living cartilage implant.
When it comes to cartilage tissue donation, allogeneic solutions (tissues from donors rather than the patient) are preferred over autologous technology (tissue from the patient) for several reasons, not the least of which is the extreme benefit of juvenile tissue. According to company CEO Mitchell Seyedin, Ph.D., cells that are younger than a year old have at least a 100-fold difference in repair and regeneration capacity. Other benefits of allogeneic tissue include the need for only one surgery instead of two; the availability of an off-the-shelf product; economies of scale benefits; and the creation of a platform technology—such as ISTO’s—that potentially can be leveraged into additional applications.
NuQu, ISTO’s cell-based technology platform for cartilage regeneration, is a minimally invasive surgical treatment for lumbar and discogenic back pain. A Phase II trial for the product began in the fourth quarter of 2012; though currently only in trial for spinal applications, Seyedin told ODT that he hopes to see it applied to knees and shoulders as well.
“Data from 15 patients treated in the Phase I trial shows considerable promise in safety, with the potential of offering patients relief with little or no recovery time,” said Domagoj Coric, M.D., lead clinical investigator for the project and a practicing neurosurgeon at Carolina Neurosurgery and Spine Associates in Charlotte, N.C. “NuQu could become a preferred alternative for those patients with chronic low back pain, who do not respond to traditional, non-surgical care such as physical therapy or epidural steroid injections.
Currently, the only options for these patients are to either live in pain or to undergo a major surgical procedure.”
ISTO’s InQu technology is a bone graft extender and substitute, and it received 510(k) clearance from the U.S. Food and Drug Administration in 2007. The technology was launched in the United States the following year, and won the Frost & Sullivan Award for Product Innovation that same year. The InQu bone graft extender and substitute combines the structural and biological attributes of polylactidecoglycolide (PLGA) and hyaluronic acid (HyA), two biomaterials. PLGA gives 3-D structure to InQu, and is meant to provide a reliable resorption rate at the site of implantation. HyA is reported to play a critical role in tissue regeneration and repair, and is known to aid essential early events in bone formation by promoting the migration, proliferation, and differentiation of osteogenic cells.
According to ASG Reports, a market research and analysis firm based in Amsterdam, The Netherlands, the global orthopedics devices market is forecast to reach a value of $61.3 billion by 2017 from a market value of $36.7 billion in 2010 at a compound annual growth rate of 7.6 percent. The report identified minimally invasive devices as being the driver behind the new technologies market. Companies such as Baxano Surgical (formerly Trans1) wisely are anticipating this trend by creating a strong foothold and a robust portfolio in the minimally invasive surgery (MIS) orthopedics market.
Finding—and creating—niche markets is becoming essential to survival for smaller medical device companies. While the large medical device companies such as DePuy Synthes, Stryker, Zimmer Holdings Inc., Smith & Nephew plc and Medtronic Inc. together hold more than 60 percent of market share in the global orthopedic devices market, smaller companies can maintain a strong foothold if they innovate and stick to what they know best. EOS Imaging, for instance, has harnessed Nobel Prize-winning technology to create a unique orthopedic imaging system that no other company really has a finger in.
“Decreasing profit margins and pressure to launch new products has led to consolidation,” read an ASG report on orthopedic market growth published in March last year. “For example, in 2011 DePuy acquired Synthes. Furthermore, increasing competition from local companies with low cost products has led to an increased focus on biomaterials and orthobiologics, meaning that these products could hold the key to growth in the orthopedic devices industry.”
Orthobiologics, which includes regenerative medicine, deals with growing new human tissue instead of using artificial implants to heal fractures and other orthopedic ailments. Companies such as ISTO Technologies and Bioventus are harnessing stem cell technology and ultrasound pulses to promote and accelerate the body healing itself.
EOS Imaging
Location: Paris, France
Leadership: Marie Meynadier, Ph.D., CEO
Sector: Orthopedic imaging
Online: www.eos-imaging.com
EOS Imaging SA is built upon a technology that won the 1992 Nobel Prize for Physics: a high energy particle detector. Georges Charpak’s invention detected “radiations that help understand the origin of the universe,” explained ESO CEO Marie Meynadier, Ph.D., to Orthopedic Design & Technology; and EOS has harnessed that capability in its unique imaging system that can take three dimensional images of human bone in weight-bearing positions, all with a very low radiation dose.
Meynadier, who has a background in microelectronics, started EOS in 2005 as a young startup by the name of Biospace Med. The company changed its name to EOS Imaging in 2010 when the EOS brand really took off and gained recognition. “Doctors started asking for ‘an EOS exam,’ not a ‘pair of X-rays,’” said Meynadier. By 2010, the EOS imaging system had become as recognizable to those using it as a CT (computed tomography) or MRI (magnetic resonance imaging) scan.
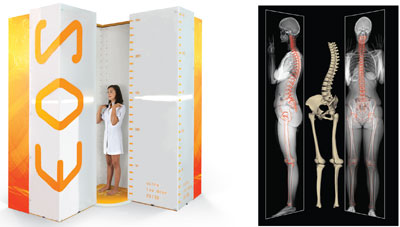
|
The EOS System scans the patient’s body while he or she is in a standing, weight-bearing position. Image courtesy of EOS Imaging SA. Full body scan image produced by the EOS System. Image courtesy of EOS Imaging SA. |
The EOS System, which EOS manufactures, provides exactly that—a set of very low dose X-rays from which 3-D imaging of the skeleton is derived. When Meynadier met Charpak, he had a groundbreaking technology that he needed to market, and Meynadier had an idea of where his particle detector could be best used. Bone imaging presents unique challenges: Physicians and medical professionals often need to view large sections of the patient’s body at once, such as whole legs or whole spines, and the radiation associated with these exams was high. Besides, traditional X-rays only give a single-angle, “flat” view. CT and MRI machines can provide 3-D imaging, but when the patient’s body is recumbent, the joints are not being viewed in a functional position. Also, CT scans place the body under high doses of radiation, while the EOS system is emits very low doses of the harmful rays.
“EOS takes two radiographs of the patient, frontal and lateral simultaneously,” explained Meynadier. “It does it at a very low dose and it has the ability to do that over the whole body of a patient. Then, with software, we can reconstruct an envelope of the patient’s bones in 3-D in a weight bearing position, and extract a full set of clinical parameters that the surgeons want, whether it’s in spine, hip or knee surgery.”
EOS’ growth has exploded in the past two years. Its revenues in 2012 were $9.14 million, 36 percent above the year before. Already for the first quarter of 2013, revenues are $2.49 million, while for the same quarter last year they were $730,000. Sixty-two percent of revenues in Q1 2013 were generated from North America, where the company sold three EOS systems. In Europe, revenues were $790,000, which included the sale of one EOS system.
2012 was a landmark year for EOS Imaging. The company gained significant financial resources from an initial public offering in February, which allowed it to make “controlled commercial investments to bolster growth in 2013 and beyond,” according to Meynadier. Last year, the company also saw EOS adopted in four of the top seven U.S. orthopedic hospitals. The United States will be a key focus region for future growth, and EOS has offices in both Cambridge, Mass., and Montreal, Canada. EOS also
recently opened an office in Singapore to serve the Asian market.
EOS manufacturing operations are located in France, and the company has no plans to move manufacturing anywhere else. “There are lots of non-quality issues linked to manufacturing everywhere on the planet,” Meynadier said. “At our size, for the years to come, we believe that our manufacturing has been strongly secured; keeping manufacturing close to our headquarters is the best option for EOS.”
EOS has no plans to work on different products in the future; rather, the company will continue to focus on expanding the capabilities of the EOS system.
“There are a large number of avenues of usage in the orthopedic practice that we are investigating with surgeon partners,” Meynadier said. “Not only with diagnostics, but also planning, simulation and quality control. We are more focused on developing more software features for EOS than developing new devices. Quality control for hip and knee surgery, for instance, is extremely straightforward with the EOS system. This is a very strong asset to an accountable care organization for its ability to plan a given surgery, execute it and control its quality with a reliable tool.”
Baxano Surgical
Location: Raleigh, N.C.
Leadership: Ken Reali, President and CEO
Sector: Degenerative conditions of lumbar spine
Online: www.baxanosurgical.com
At the end of May this year, Trans1 Inc. completed the acquisition of Baxano Inc. and became
Baxano Surgical Inc. The newly branded company describes itself as a medical device company focused on products to treat degenerative conditions of the spine affecting the lumbar region. Baxano Surgical currently markets the AxiaLIF family of products for single and two level lumbar fusion, the VEO lateral access and interbody fusion system, and the iO-Flex system, a proprietary minimally invasive set of flexible instruments allowing surgeons to target lumbar spinal stenosis during spinal decompression procedures in all three regions of the spine: central canal, lateral recess, and neural foramen.
The merger was part of a four-part plan that was conceived in 2010 when Ken Reali joined Trans1 as CEO. First, the company needed to cut its burn rate and work towards establishing positive cash flow. To help this goal, Trans1 needed to establish physician reimbursement for AxiaLIF, which it did Jan. 1 this year. Thirdly, the company launched Veo early last year. The final part of the plan was an acquisition.
“We’ve had our eye on Baxano since 2010,” Reali told ODT. “We really liked the iO-Flex technology and we saw a direct fit and synergy with our fusion systems AxiaLIF and Veo. The acquisition not only gives us a timeline to get to profitability, but shortens our timeline to profitability by a year.”
The company was founded as Trans1 in 2000, when Andy Cragg, M.D., an interventional radiologist in Minneapolis, Minn., had the idea for a pre-sacral approach to the lumbar spine. Cragg’s brother, an orthopedic surgeon, observed that the damage he would do to the soft tissue structures through traditional access to the lumbar spine seemed to cause more pain to the patient than their original back pain. Recovery periods were long and this gave lumbar fusion surgery a bad reputation. Cragg’s own experience in interventional radiology, which is concerned with using minimally invasive image-guided procedures to diagnose and treat diseases in nearly every organ system, influenced him to consider the spinal column as a tubular structure. With this vision, he developed a method of accessing the lumbar spine via the spine’s axis rather than perpendicularly from the side, front, or back. Cragg then solicited the help of engineer Bob Assell who took the physician’s catheter thinking and turned it into proprietary instrumentation that accomplished the task while retaining a design familiar to orthopedic and neuro surgeons.
This instrumentation eventually became Baxano’s AxiaLIF system: Nitinol was used in the construction of special cutters designed to debulk the nucleus of the disc and rough up the vertebral endplates; and stainless steel tissue extractors were created to remove the disc material so that bone graft could be inserted into the disc space. In 2004, AxiaLIF earned 510(k) clearance from the U.S. Food and Drug Administration (FDA). The very next year, Baxano Inc. was founded. Baxano was founded by Jeff Bleich, M.D., a physician at Stanford University Medical Center, who had recognized the significant clinical need for a less invasive, less traumatic means of achieving a lumbar spinal decompression.
Together with serialentrepreneur Vahid Saadat, Bleich led a team of clinical advisors and engineers to develop a set of modified rongeurs (instruments with a sharp-edged, scoop-shaped tip, used for gouging out bone) that would become the first iO-Flex prototypes.
In 2007, Baxano received FDA clearance to market the iO-Flex Microblade Shaver. The company gained a U.S. patent on the system in 2009, and named Tony Recupero CEO for his extensive experience in sales the same year. Baxano was focused on aggressively expanding its intellectual property portfolio, and Recupero was the man for the job. Last year, iO-Flex earned CE mark and Health Canada approval to market in European Union countries and Canada.
“The synergies of multiple lines of differentiated MIS technologies, serving specialists within spine surgery focusing on MIS makes strategic sense to us, and the deal also scales up the growth model significantly for the company over the next two to three years,” wrote Piper Jaffray analyst Matt Miksic upon news of the merger. “In addition to its strategic positives, the financing associated with the deal will delay (but not eliminate) the need to raise additional cash, easing an overhang on TranS1 since January.”
The future of Baxano Surgical is all about new products. The company’s new minimally invasive facetectomy removal instrument, iO-Tome, was cleared by the FDA in October last year, and Baxano plans to release it in the fourth quarter of 2013. The instrument is designed to rapidly and precisely remove the facet joint, a procedure commonly required in spinal fusion applications. In the pipeline for next year is a minimally invasive pedicle screw system called Avance.
“We will exit this year with four flagship products, all proprietary and differentiated, and all focused in the minimally invasive spine arena,” said Reali.
Bioventus
Location: Durham N.C.
Leadership: Mark Augusti, CEO
Sector: Orthobiologics
Online: www.bioventusglobal.com
Bioventus LLC is a very young company, having been founded just last year. However, the company spun off from the Biologics and Clinical Therapies division of one of the largest orthopedic companies in the world, Smith & Nephew, so it was born with a considerable advantage. The spinoff was a strategic venture between an Essex Woodlands-led investor syndicate group and Smith & Nephew.
The company’s flagship product is Exogen, a system that uses ultrasound signals to accelerate bone healing. It is the only U.S. Food and Drug Administration-approved device that uses a low intensity pulsed ultrasound signal for bone healing. It is indicated for use in non-union fractures (fractures that have not healed properly) and certain kinds of stress fractures.
Bioventus also markets Supartz in the United States. This is a three- to five-injection regimen for the treatment of mild to moderate osteoarthritis pain in patients who do not respond to steroids or analgesics. Outside the United States, the company markets Durolane, which is a single injection technology for the same indication. The company intends to bring Durolane stateside, and is working with U.S. regulatory authorities to begin clinical trials and the process for approval of the product.
The products Bioventus makes fall into the category of, as CEO Mark Augusti describes it, “active orthopedic healing.” This kind of technology, Augusti explained last year to ODT, is often described as orthobiologics. Studies show, however, that this type of healing has a sociological effect, which is why Bioventus brands its approach a little differently.
Upon its founding, Bioventus was still operating under the name Smith & Nephew outside the United States. Smith & Nephew was working essentially as the company’s distributor; changing the branding would happen gradually as Bioventus navigated various countries’ labor regulations. In December last year, Bioventus opened its international headquarters in The Netherlands just outside Amsterdam, which enabled Bioventus’ products distributed there to be transitioned to the Bioventus brand. Italy was next to receive the Bioventus brand and products; then the United Kingdom, Ireland, Sweden, Norway, Denmark, Canada, France, Finland, and most recently, Germany.
When Augusti took the reigns as CEO of Bioventus at its inception, he was already a seven-year veteran of Smith & Nephew, and was serving as president of the company’s Biologics & Spine business. Key appointments since then have been David Price as chief financial officer, who took on the role in October last year; and Peter Heeckt, M.D., Ph.D., as chief medical officer, appointed in April this year. Price joined Bioventus from EDGAR Online Inc., a financial data, technology and business process outsourcing company; and Heeckt most recently was co-founder and managing member for Life Sciences Consulting Services LLC, a consulting firm specializing in medical and clinical affairs and healthcare compliance for medical device and biotech companies.
Bioventus is earning more global street cred as its products become recognized by international regulatory and other bodies. In May this year, the NHS (National Health Service) Technology Adoption Centre in the United Kingdom, the implementation arm of the National Institute for Health and Care Excellence (NICE), recently launched a new adoption pack regarding Exogen. The adoption pack is designed to help NHS trusts correctly implement guidelines NICE released supporting the use of Exogen as an alternative to surgery for thousands of patients with long bone non-union fractures. The pack includes information about the benefits of Exogen to patients, physicians and healthcare systems, the national and local opportunities available, the drivers for adoption, detailed case studies of where Exogen has already been successfully used in the NHS, and the costs and potential savings.
ISTO Technologies
Location: St. Louis, Mo.
Leadership: Mitchell Seyedin, Ph.D., President and CEO
Sector: Orthobiologics
Online: www.istotech.com
Regenerative medicine is an exciting new field in orthopedics, not least because of its challenges. Vague laws and very young research make the field of tissue regeneration difficult to navigate, but dynamic St. Louis, Mo.-based orthobiologics company ISTO Technologies Inc. has been working since 2002 to perfect cartilage regeneration, which is particularly problematic. Finding stem cells that can regenerate cartilage reliably without fusing with and turning into hard bone down the line has proved very difficult. Recent research has shown that children younger than one year of age have much more of a cell population capable of regenerating cartilage than anyone else. ISTO holds patents on juvenile cell-based technology that they use in their products.
ISTO began operations in 2002 by sharing the development responsibilities of Denovo ET (engineered tissue) with Zimmer Holdings. This year, just over a decade later, ISTO took full control of the technology. Denovo ET is an engineered cartilage implant designed to repair defects in the knee.
ISTO renamed the product Revaflexand currently is proceeding independently with further development of the program through a Phase III clinical program. The technology harnesses juvenile cartilage cells to create a living cartilage implant.
When it comes to cartilage tissue donation, allogeneic solutions (tissues from donors rather than the patient) are preferred over autologous technology (tissue from the patient) for several reasons, not the least of which is the extreme benefit of juvenile tissue. According to company CEO Mitchell Seyedin, Ph.D., cells that are younger than a year old have at least a 100-fold difference in repair and regeneration capacity. Other benefits of allogeneic tissue include the need for only one surgery instead of two; the availability of an off-the-shelf product; economies of scale benefits; and the creation of a platform technology—such as ISTO’s—that potentially can be leveraged into additional applications.
NuQu, ISTO’s cell-based technology platform for cartilage regeneration, is a minimally invasive surgical treatment for lumbar and discogenic back pain. A Phase II trial for the product began in the fourth quarter of 2012; though currently only in trial for spinal applications, Seyedin told ODT that he hopes to see it applied to knees and shoulders as well.
“Data from 15 patients treated in the Phase I trial shows considerable promise in safety, with the potential of offering patients relief with little or no recovery time,” said Domagoj Coric, M.D., lead clinical investigator for the project and a practicing neurosurgeon at Carolina Neurosurgery and Spine Associates in Charlotte, N.C. “NuQu could become a preferred alternative for those patients with chronic low back pain, who do not respond to traditional, non-surgical care such as physical therapy or epidural steroid injections.
Currently, the only options for these patients are to either live in pain or to undergo a major surgical procedure.”
ISTO’s InQu technology is a bone graft extender and substitute, and it received 510(k) clearance from the U.S. Food and Drug Administration in 2007. The technology was launched in the United States the following year, and won the Frost & Sullivan Award for Product Innovation that same year. The InQu bone graft extender and substitute combines the structural and biological attributes of polylactidecoglycolide (PLGA) and hyaluronic acid (HyA), two biomaterials. PLGA gives 3-D structure to InQu, and is meant to provide a reliable resorption rate at the site of implantation. HyA is reported to play a critical role in tissue regeneration and repair, and is known to aid essential early events in bone formation by promoting the migration, proliferation, and differentiation of osteogenic cells.
|
By Definition... According to the National Institutes of Health, regenerative medicine is the process of replacing or regenerating human cells, tissues ororgans to restore or establish normal function. This field holds the promise of regenerating damaged tissues and organs in the body by replacing damaged tissue and/or by stimulating the body’s own repair mechanisms to heal previously irreparabletissues or organs. |