David C. Kelman, President, Kelman & Company Consulting LLC03.21.18
Developing new products is not an easy task to accomplish. It takes a tremendous amount of creativity, fortitude, and sheer dogged determination. And that is just to travel from concept to a final form for commercial sale. Then the true gut wrenching occurs. Do the potential customers value the new and creative solutions a company’s engineers have toiled over, defended, and fought for to address specific market needs such that they will stop using what they have been and try the new product instead? The greater the commercial success, the more one realizes the long, hard hours were worth it. Then there were instances when all the requirements were solved, but the commercial success did not materialize, or at least not to the extent an organization had planned. Why? The process followed has been tried and true. So why the inconsistent results?
In 1998, Doblin1 identified 10 types of innovation (Figure 1), and for an innovation to be successful, it required a minimum of three to four types of innovation.
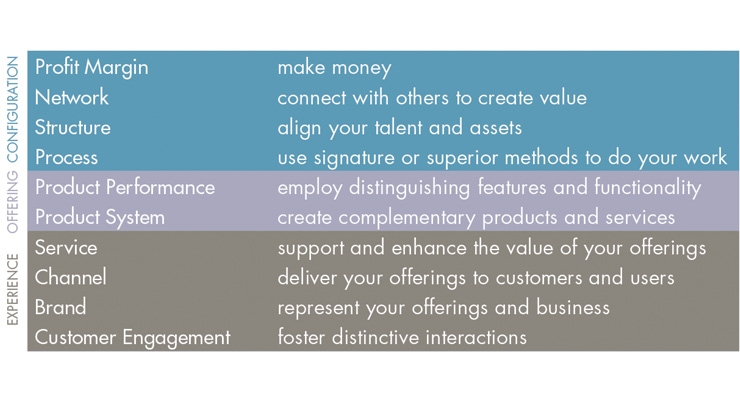
Figure 1: Doblin Ten Types of Innovation
Looking back at the commercial product “failures” I had experience with, I can now see that there was only one type of innovation utilized. That was Product Performance (some may refer to it as “features and benefits”). In these situations, there was an existing competitive product that was commercially successful. The requirement for the product development was to develop a very similar product with a few additional features and benefits.
If examined from the perspective of total joint innovation over the last several decades, it is easy to see how continued innovation in this space has changed. In the simplest terms, someone has an issue or a problem with what is currently “state of the art.” An example of this is Sir John Charnley and his innovation of the low friction hip arthroplasty. The low friction arthroplasty offered an innovative solution, but it importantly focused on the surgeon’s experience as well. A complete offering was developed to address the need of patients with severe pain, who could not sleep, and had very limited mobility. The solution eliminated the pain and provided basic mobility from bed to bath. There were easily eight different types of innovation involved in what Sir Charnley created. All the various types of innovations were necessary for success.
Since then, continued innovation has occurred as new opportunities were identified, again employing multiple types of innovations, which has expanded the availability to patients younger than Charnley’s age restriction of 65 and older. Approximately 43 percent of the total joint patients today are under the age of 65 and seek the ability to return to their active lifestyles versus gaining pain relief with limited mobility. Examining the types of innovations identified by Doblin, such as Product System and Customer Engagement, we can see how this area has changed over the years. Early on, a “knee system” was either a cruciate retaining knee or a posterior stabilized. The evolution of that knee system today is a comprehensive solution, comprised of components including cruciate retaining, posterior stabilized, and revision arthroplasty, coupled with the ability to easily move between the various components. The Customer Experience has evolved with enhanced instrumentation, patient specific instrumentation, and numerous education venues. These innovations have enabled the surgical technique to be reproducible.
Total joint replacement is in the maturity aspect of the technology curve (Figure 2). At this point, it is common for innovation to be incremental and to have strong competition between competitors. In a mature market, there is focus on efficiency and cost, while also incorporating multiple types of innovations.
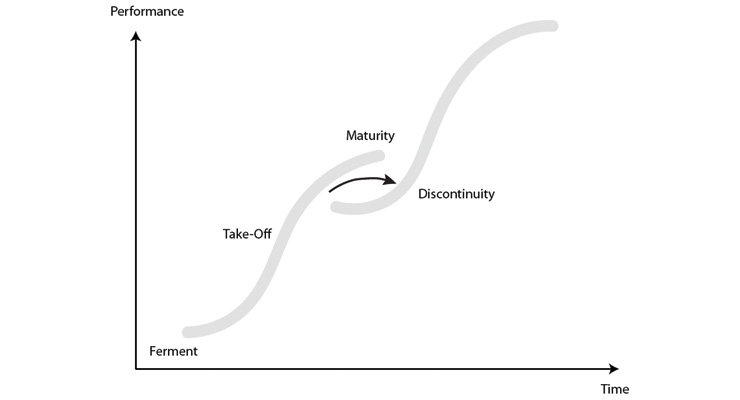
Figure 2: Technology S Curve of Maturity2
Additionally, the burden of the high cost of healthcare on the national economy adds even more pressure to reduce the cost. Healthcare costs as a percentage of gross domestic product has continued to rise from approximately 7 percent in 1970 to almost 18 percent today3 (Figure 3).
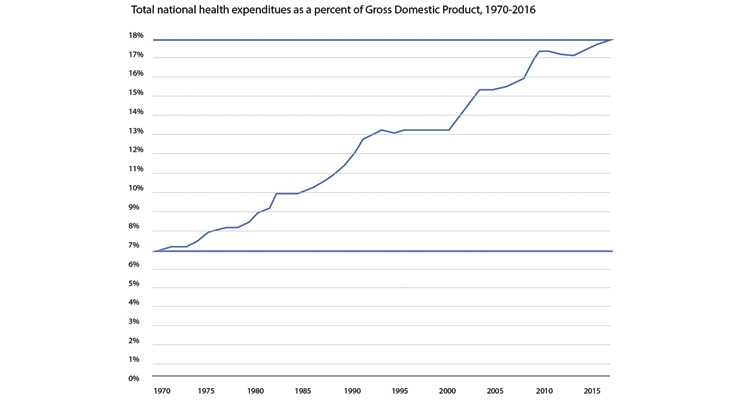
Figure 3: Health spending growth has outpaced growth of the U.S. economy.3
Regardless of what happens (or does not happen) in Washington D.C., the pressure regarding the costs of U.S. healthcare will continue.
Future development—regardless if it is in the early stage of the technology curve or more in the maturity aspect of the curve—is no longer simply about improving patient outcomes. But how does it improve patient outcomes while reducing costs? Cost is a very ambiguous word. Some of the easiest views are the cost to produce the product, or the purchase price, but it is much more complicated than those simple views. For the medical device manufacturer, there is a need to consider a more holistic cost, including everyone’s time involved in getting the product to the OR, actual product costs, shipping costs, etc. From the hospital’s perspective, cost to sterilize, storage space requirements (cost/square foot), staff training cost, etc. The costs list goes on. All of these offer the potential for future innovations as well as continued product and system performance improvements.
The job of the development engineer is even more difficult today. Not only must a new product be an innovative concept, but it must provide financial benefits to all stakeholders, and also yield better outcomes for the patient. No longer will the rationale that “10 sizes are better than the competitor’s eight sizes” be sufficient, unless there are clinical outcomes and financial benefits incorporated. As the development process progresses, routine cost impact analyses will need to be performed just as 3D printed prototypes are produced throughout a development process. If a new feature or additional capability is to be incorporated, it must be determined what the impact to the entire product cost is—again from the point of view of all stakeholders.
To be as commercially successful as possible, remember it is necessary to utilize at least three to four different types of innovation, and the more implemented, the greater the possible commercial success. As the industry moves from a fee-for-service model to a value-based payment model, surgeons are much more concerned with the cost of a procedure. Cost benefits must be considered and proven, regardless if a project is an emerging technology or an incremental improvement.
References
David C. Kelman, president of Kelman & Company Consulting LLC, is a licensed professional engineer who has managed and lead design and development teams on three different continents for major orthopedic companies. He is a creative innovator whose broad scope of solutions has resulted in two unique start-up businesses, commercialization of 40 product platforms, and 50 U.S. patents awarded. As a multifaceted problem solver, he incorporates technological and business sophistication across diverse environments, including product design/software/analytics/supply chain/technical marketing and sales. This unique mix of technical, engineering, and business expertise has resulted in successful business strategies, and the introduction of cutting-edge technologies with financial viability.
In 1998, Doblin1 identified 10 types of innovation (Figure 1), and for an innovation to be successful, it required a minimum of three to four types of innovation.

Figure 1: Doblin Ten Types of Innovation
Looking back at the commercial product “failures” I had experience with, I can now see that there was only one type of innovation utilized. That was Product Performance (some may refer to it as “features and benefits”). In these situations, there was an existing competitive product that was commercially successful. The requirement for the product development was to develop a very similar product with a few additional features and benefits.
If examined from the perspective of total joint innovation over the last several decades, it is easy to see how continued innovation in this space has changed. In the simplest terms, someone has an issue or a problem with what is currently “state of the art.” An example of this is Sir John Charnley and his innovation of the low friction hip arthroplasty. The low friction arthroplasty offered an innovative solution, but it importantly focused on the surgeon’s experience as well. A complete offering was developed to address the need of patients with severe pain, who could not sleep, and had very limited mobility. The solution eliminated the pain and provided basic mobility from bed to bath. There were easily eight different types of innovation involved in what Sir Charnley created. All the various types of innovations were necessary for success.
Since then, continued innovation has occurred as new opportunities were identified, again employing multiple types of innovations, which has expanded the availability to patients younger than Charnley’s age restriction of 65 and older. Approximately 43 percent of the total joint patients today are under the age of 65 and seek the ability to return to their active lifestyles versus gaining pain relief with limited mobility. Examining the types of innovations identified by Doblin, such as Product System and Customer Engagement, we can see how this area has changed over the years. Early on, a “knee system” was either a cruciate retaining knee or a posterior stabilized. The evolution of that knee system today is a comprehensive solution, comprised of components including cruciate retaining, posterior stabilized, and revision arthroplasty, coupled with the ability to easily move between the various components. The Customer Experience has evolved with enhanced instrumentation, patient specific instrumentation, and numerous education venues. These innovations have enabled the surgical technique to be reproducible.
Total joint replacement is in the maturity aspect of the technology curve (Figure 2). At this point, it is common for innovation to be incremental and to have strong competition between competitors. In a mature market, there is focus on efficiency and cost, while also incorporating multiple types of innovations.

Figure 2: Technology S Curve of Maturity2
Additionally, the burden of the high cost of healthcare on the national economy adds even more pressure to reduce the cost. Healthcare costs as a percentage of gross domestic product has continued to rise from approximately 7 percent in 1970 to almost 18 percent today3 (Figure 3).

Figure 3: Health spending growth has outpaced growth of the U.S. economy.3
Regardless of what happens (or does not happen) in Washington D.C., the pressure regarding the costs of U.S. healthcare will continue.
Future development—regardless if it is in the early stage of the technology curve or more in the maturity aspect of the curve—is no longer simply about improving patient outcomes. But how does it improve patient outcomes while reducing costs? Cost is a very ambiguous word. Some of the easiest views are the cost to produce the product, or the purchase price, but it is much more complicated than those simple views. For the medical device manufacturer, there is a need to consider a more holistic cost, including everyone’s time involved in getting the product to the OR, actual product costs, shipping costs, etc. From the hospital’s perspective, cost to sterilize, storage space requirements (cost/square foot), staff training cost, etc. The costs list goes on. All of these offer the potential for future innovations as well as continued product and system performance improvements.
The job of the development engineer is even more difficult today. Not only must a new product be an innovative concept, but it must provide financial benefits to all stakeholders, and also yield better outcomes for the patient. No longer will the rationale that “10 sizes are better than the competitor’s eight sizes” be sufficient, unless there are clinical outcomes and financial benefits incorporated. As the development process progresses, routine cost impact analyses will need to be performed just as 3D printed prototypes are produced throughout a development process. If a new feature or additional capability is to be incorporated, it must be determined what the impact to the entire product cost is—again from the point of view of all stakeholders.
To be as commercially successful as possible, remember it is necessary to utilize at least three to four different types of innovation, and the more implemented, the greater the possible commercial success. As the industry moves from a fee-for-service model to a value-based payment model, surgeons are much more concerned with the cost of a procedure. Cost benefits must be considered and proven, regardless if a project is an emerging technology or an incremental improvement.
References
- Ten Types of Innovation: The Discipline of Building Breakthroughs, November 20, 2014. http://bit.ly/odt180450
- The Innovation S-Curve, Gal’s Insights, July 25, 2015. http://bit.ly/odt180451
- http://bit.ly/odt180452
David C. Kelman, president of Kelman & Company Consulting LLC, is a licensed professional engineer who has managed and lead design and development teams on three different continents for major orthopedic companies. He is a creative innovator whose broad scope of solutions has resulted in two unique start-up businesses, commercialization of 40 product platforms, and 50 U.S. patents awarded. As a multifaceted problem solver, he incorporates technological and business sophistication across diverse environments, including product design/software/analytics/supply chain/technical marketing and sales. This unique mix of technical, engineering, and business expertise has resulted in successful business strategies, and the introduction of cutting-edge technologies with financial viability.