By Sean Fenske, Editor-in-Chief
While the pandemic put a pause on the controversy surrounding EtO sterilization and any action taking place by the EPA or FDA, the matter is far from resolved. In fact, a number of medical device manufacturers have used the additional time to explore other sterilization options for products still in development. As such, there’s perhaps never been more uncertainty over medical device sterilization or what process to use.
Providing a solution of its own, De Lama has developed its HyPerPure® technology, which leverages hydrogen peroxide, but has coupled it with a unique process and equipment to enable it to be used for a broader spectrum of products. This low-temperature sterilization process can be used for a number of medical devices that were not previously able to be treated with hydrogen peroxide.
To fully explain this technology, Marco Bianchi, marketing and sales manager, and Guido Rovera, marketing & sales director, of De Lama spa, took time to explain the benefits of their system, how it differs from traditional hydrogen peroxide sterilization methods, and how it compares to EtO.
Sean Fenske: Can you please explain why it’s important for companies to explore medical device sterilization alternatives outside of ethylene oxide (EtO)?
Marco Bianchi: Our company has been producing EtO sterilizers for more than 50 years; this equipment is still available in the De Lama catalog and continues to be delivered today. This technology, however, has always proved challenging to handle—the media is toxic, carcinogenic, and explosive. These problems make it expensive and difficult to manage. In addition, international regulations have gotten more and more restrictive. As a result, FDA recently promoted the development of alternative sterilization methods.
Considering the shortcomings of EtO, the FDA’s call to action, and urging from customers, De Lama took on the challenge to develop an alternative sterilization method. Our HyPerPure® technology represents the success of those efforts.
Fenske: What is HyPerPure® with vapor phase hydrogen peroxide and how does it differ from other hydrogen sterilization technology?
Guido Rovera: Traditionally, hydrogen peroxide technology is associated with superficial sterilization, for which a better name would be “decontamination.” With traditional hydrogen peroxide technology, media is vaporized, and as a sterilizing agent, ensures its action by aspersion on only the visible surface of the product. With a simple vaporization, it is not possible to penetrate complex product geometries or Tyvek® packaging, where normally medical devices and prostheses are positioned before sterilization with EtO or gamma rays.
De Lama’s HyPerPure® technology differs from “traditional” hydrogen peroxide decontamination, because it works under deep vacuum and zero-air condition. With this method, hydrogen peroxide is able to reach all internal surfaces, just as what occurs with EtO. For example, we can penetrate and sterilize the internal part of long, ultra-thin catheters, something that is absolutely impossible with traditional hydrogen peroxide techniques. We are also able to penetrate Tyvek® packaging and we ensure a complete sterilization (12-log) of all products contained inside the packaging.
In a way, we reproduce what happens with traditional saturated steam sterilization, where air in the chamber is totally replaced by steam in a saturated state.
Additionally, De Lama's HyPerPure® generator performs ionization of vaporized H202, improving the sterilization efficacy.
The extended firsthand experience of De Lama in the production of saturated steam sterilizers, EtO sterilizers, and vacuum dryers during the last 70 years drove the De Lama Technical Department through the development of this new technology. This gave us a big advantage in the study and development of this kind of process.

De Lama HyPerPure® equipment sizes range from small laboratory-sized equipment of 500-liter chambers up to huge production facility options with 50-cubic meters (or more) of capacity.
Fenske: Can you also explain how this compares to plasma H2020 sterilization?
Rovera: Yes, we wanted to also touch on this point. To make the overall picture complete, we must also mention another “traditional” technique where hydrogen peroxide (H202) is involved—plasma H202 sterilization. In this case, H202 media is used with a cartridge and an electric field is generated to disaggregate H202 molecules. In some cases, a vacuum (not deep vacuum) is used.
This technology is more effective than simple H202 vaporization but has a significant limitation: it works with an electric field that can only be created in small-sized equipment. This equipment is used in hospitals to sterilize small surgical tools or other medical tools, but as the chamber size can never be bigger than 400 liters, only a few products can be treated at a time. You can easily understand why this technology can't be used within manufacturing and production facilities, while De Lama HyPerPure® equipment sizes range from small laboratory-sized equipment of 500-liter chambers up to huge production facility options with 50-cubic meters (or more) of capacity. De Lama equipment doesn’t need an electric field to disaggregate H202 molecules as we use a different method. In addition to this, a plasma sterilizer can't treat products packaged inside Tyvek® or medical paper, while this is a plus for De Lama's HyPerPure® equipment.
Another difference from plasma H2020 sterilizers is the fact De Lama's HyPerPure®, rather than expensive cartridges, uses the “classic” 35% concentration hydrogen peroxide for commercial use, which is readily available on the general market in plastic tanks.
Fenske: Are there any environmental and/or safety issues with HyPerPure® such as the concerns surrounding EtO sterilization?
Rovera: As mentioned previously, EtO is toxic, explosive, carcinogenic, and difficult to handle. HyPerPure® works only with hydrogen peroxide media, which, thanks to the De Lama process, is destroyed at the end of the cycle through a catalyst, which disaggregates H202 into hydrogen and oxygen. After this step, all the molecules can be released into the atmosphere in a totally eco-friendly way.
H202 is not explosive and is easy to store in plastic tanks without any need for special handling or storage. De Lama equipment doors can be opened at the end of the cycle when 1 ppm concentration is reached, as regulations require. Also, as a reminder, the H202 is the same liquid present in many homes, used for disinfection of minor wounds.

A game-changing advantage for many manufacturers is the fact the HyPerPure® equipment can be installed directly inside a production facility.
Fenske: What other benefits does HyPerPure® offer as compared to EtO?
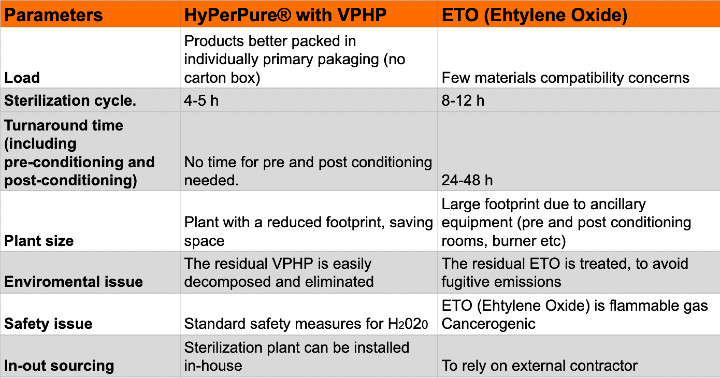
Bianchi: First, the HyPerPure® sterilization process has an average duration of three to five hours versus the eight to 12 hours for EtO. Further, use of EtO requires additional preconditioning and postconditioning processes, bringing the overall duration to closer to 24 to 48 hours. A significant difference when compared to three to five hours to accomplish the same result with HyPerPure®. Given this difference, it’s easy to see how the production rate can dramatically increase as a result.
In addition, there is a substantial space (and capital expenditures) savings as all ancillary equipment required for EtO are no longer needed. This includes burners, preconditioning and postconditioning rooms, store rooms for cylinder refill, etc.
A game-changing advantage for many manufacturers is the fact the HyPerPure® equipment can be installed directly inside a production facility. This enables further time and cost savings, as external sterilization is no longer required. It also means the company has more control of the overall product development process, with this step taking place in-house.
Fenske: What types of medical devices can be sterilized using HyPerPure® technology?
Bianchi: HyPerPure® was born out of the idea to provide a low-temperature sterilization technology; as such, all thermo-sensitive medical devices and prostheses can be treated—catheters, intracardiac catheters, syringes, cochlear implants, prostheses for implants, etc. Of course, non-thermo-sensitive products can also be treated, making the equipment ultra-flexible and productive.
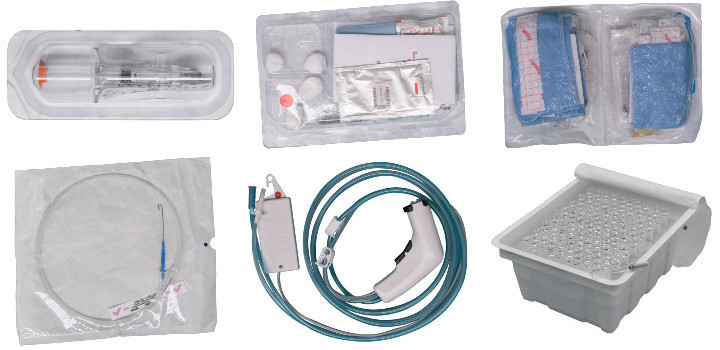
Examples of medical devices that can be sterilized with the HyPerPure® technology.
Fenske: What benefits does HyPerPure® offer from a cost standpoint? For example, what’s the impact on facility systems such as HVAC filters or the costs involved with maintenance?
Rovera: As mentioned previously, there are many advantages from a cost standpoint, but one point I want to express is the low cost of the media. As the cycle consumption of H202 is very low, so too is the cost of media compared with EtO for a single cycle—approximately 80% lower. Extrapolating that out for an entire production year, it’s easy to see how significant the savings are.
In addition, the equipment does not require any consumables; the catalyst is static. Further, there are no filters to be changed, reducing maintenance and consumables cost.
Finally, with regard to a facility’s HVAC systems, there is no impact, and the equipment can be added inside an existing plant without a specific preliminary study of air-flows.
Fenske: Do you have any additional comments you’d like to share based on any of the topics we discussed or something you’d like to tell orthopedic device manufacturers?
Rovera: Yes. We want to underline the fact HyPerPure® is not a prototype or a technology that’s still in development. Rather, it is currently available and has already been put into operation by major international companies within the pharmaceutical, medical devices, and orthopedic industries.
The process is validated and supported by existing regulations. As such, there are currently no limits to the incorporation of this technology, which represents the future of low-temperature sterilization. Presenting with ecological advantages and linked to reductions in production times and costs, HyPerPure® is a must-have for orthopedic device manufacturers.
De Lama laboratories are always open to run trials for customers specific products to verify complete sterilization of their devices as well as the compatibility of materials.
Click here to find out more about De Lama >>>>>
While the pandemic put a pause on the controversy surrounding EtO sterilization and any action taking place by the EPA or FDA, the matter is far from resolved. In fact, a number of medical device manufacturers have used the additional time to explore other sterilization options for products still in development. As such, there’s perhaps never been more uncertainty over medical device sterilization or what process to use.
Providing a solution of its own, De Lama has developed its HyPerPure® technology, which leverages hydrogen peroxide, but has coupled it with a unique process and equipment to enable it to be used for a broader spectrum of products. This low-temperature sterilization process can be used for a number of medical devices that were not previously able to be treated with hydrogen peroxide.
To fully explain this technology, Marco Bianchi, marketing and sales manager, and Guido Rovera, marketing & sales director, of De Lama spa, took time to explain the benefits of their system, how it differs from traditional hydrogen peroxide sterilization methods, and how it compares to EtO.
Sean Fenske: Can you please explain why it’s important for companies to explore medical device sterilization alternatives outside of ethylene oxide (EtO)?
Marco Bianchi: Our company has been producing EtO sterilizers for more than 50 years; this equipment is still available in the De Lama catalog and continues to be delivered today. This technology, however, has always proved challenging to handle—the media is toxic, carcinogenic, and explosive. These problems make it expensive and difficult to manage. In addition, international regulations have gotten more and more restrictive. As a result, FDA recently promoted the development of alternative sterilization methods.
Considering the shortcomings of EtO, the FDA’s call to action, and urging from customers, De Lama took on the challenge to develop an alternative sterilization method. Our HyPerPure® technology represents the success of those efforts.
Fenske: What is HyPerPure® with vapor phase hydrogen peroxide and how does it differ from other hydrogen sterilization technology?
Guido Rovera: Traditionally, hydrogen peroxide technology is associated with superficial sterilization, for which a better name would be “decontamination.” With traditional hydrogen peroxide technology, media is vaporized, and as a sterilizing agent, ensures its action by aspersion on only the visible surface of the product. With a simple vaporization, it is not possible to penetrate complex product geometries or Tyvek® packaging, where normally medical devices and prostheses are positioned before sterilization with EtO or gamma rays.
De Lama’s HyPerPure® technology differs from “traditional” hydrogen peroxide decontamination, because it works under deep vacuum and zero-air condition. With this method, hydrogen peroxide is able to reach all internal surfaces, just as what occurs with EtO. For example, we can penetrate and sterilize the internal part of long, ultra-thin catheters, something that is absolutely impossible with traditional hydrogen peroxide techniques. We are also able to penetrate Tyvek® packaging and we ensure a complete sterilization (12-log) of all products contained inside the packaging.
In a way, we reproduce what happens with traditional saturated steam sterilization, where air in the chamber is totally replaced by steam in a saturated state.
Additionally, De Lama's HyPerPure® generator performs ionization of vaporized H202, improving the sterilization efficacy.
The extended firsthand experience of De Lama in the production of saturated steam sterilizers, EtO sterilizers, and vacuum dryers during the last 70 years drove the De Lama Technical Department through the development of this new technology. This gave us a big advantage in the study and development of this kind of process.

De Lama HyPerPure® equipment sizes range from small laboratory-sized equipment of 500-liter chambers up to huge production facility options with 50-cubic meters (or more) of capacity.
Fenske: Can you also explain how this compares to plasma H2020 sterilization?
Rovera: Yes, we wanted to also touch on this point. To make the overall picture complete, we must also mention another “traditional” technique where hydrogen peroxide (H202) is involved—plasma H202 sterilization. In this case, H202 media is used with a cartridge and an electric field is generated to disaggregate H202 molecules. In some cases, a vacuum (not deep vacuum) is used.
This technology is more effective than simple H202 vaporization but has a significant limitation: it works with an electric field that can only be created in small-sized equipment. This equipment is used in hospitals to sterilize small surgical tools or other medical tools, but as the chamber size can never be bigger than 400 liters, only a few products can be treated at a time. You can easily understand why this technology can't be used within manufacturing and production facilities, while De Lama HyPerPure® equipment sizes range from small laboratory-sized equipment of 500-liter chambers up to huge production facility options with 50-cubic meters (or more) of capacity. De Lama equipment doesn’t need an electric field to disaggregate H202 molecules as we use a different method. In addition to this, a plasma sterilizer can't treat products packaged inside Tyvek® or medical paper, while this is a plus for De Lama's HyPerPure® equipment.
Another difference from plasma H2020 sterilizers is the fact De Lama's HyPerPure®, rather than expensive cartridges, uses the “classic” 35% concentration hydrogen peroxide for commercial use, which is readily available on the general market in plastic tanks.
Fenske: Are there any environmental and/or safety issues with HyPerPure® such as the concerns surrounding EtO sterilization?
Rovera: As mentioned previously, EtO is toxic, explosive, carcinogenic, and difficult to handle. HyPerPure® works only with hydrogen peroxide media, which, thanks to the De Lama process, is destroyed at the end of the cycle through a catalyst, which disaggregates H202 into hydrogen and oxygen. After this step, all the molecules can be released into the atmosphere in a totally eco-friendly way.
H202 is not explosive and is easy to store in plastic tanks without any need for special handling or storage. De Lama equipment doors can be opened at the end of the cycle when 1 ppm concentration is reached, as regulations require. Also, as a reminder, the H202 is the same liquid present in many homes, used for disinfection of minor wounds.

A game-changing advantage for many manufacturers is the fact the HyPerPure® equipment can be installed directly inside a production facility.
Fenske: What other benefits does HyPerPure® offer as compared to EtO?
Bianchi: First, the HyPerPure® sterilization process has an average duration of three to five hours versus the eight to 12 hours for EtO. Further, use of EtO requires additional preconditioning and postconditioning processes, bringing the overall duration to closer to 24 to 48 hours. A significant difference when compared to three to five hours to accomplish the same result with HyPerPure®. Given this difference, it’s easy to see how the production rate can dramatically increase as a result.
In addition, there is a substantial space (and capital expenditures) savings as all ancillary equipment required for EtO are no longer needed. This includes burners, preconditioning and postconditioning rooms, store rooms for cylinder refill, etc.
A game-changing advantage for many manufacturers is the fact the HyPerPure® equipment can be installed directly inside a production facility. This enables further time and cost savings, as external sterilization is no longer required. It also means the company has more control of the overall product development process, with this step taking place in-house.
Fenske: What types of medical devices can be sterilized using HyPerPure® technology?
Bianchi: HyPerPure® was born out of the idea to provide a low-temperature sterilization technology; as such, all thermo-sensitive medical devices and prostheses can be treated—catheters, intracardiac catheters, syringes, cochlear implants, prostheses for implants, etc. Of course, non-thermo-sensitive products can also be treated, making the equipment ultra-flexible and productive.

Examples of medical devices that can be sterilized with the HyPerPure® technology.
Fenske: What benefits does HyPerPure® offer from a cost standpoint? For example, what’s the impact on facility systems such as HVAC filters or the costs involved with maintenance?
Rovera: As mentioned previously, there are many advantages from a cost standpoint, but one point I want to express is the low cost of the media. As the cycle consumption of H202 is very low, so too is the cost of media compared with EtO for a single cycle—approximately 80% lower. Extrapolating that out for an entire production year, it’s easy to see how significant the savings are.
In addition, the equipment does not require any consumables; the catalyst is static. Further, there are no filters to be changed, reducing maintenance and consumables cost.
Finally, with regard to a facility’s HVAC systems, there is no impact, and the equipment can be added inside an existing plant without a specific preliminary study of air-flows.
Fenske: Do you have any additional comments you’d like to share based on any of the topics we discussed or something you’d like to tell orthopedic device manufacturers?
Rovera: Yes. We want to underline the fact HyPerPure® is not a prototype or a technology that’s still in development. Rather, it is currently available and has already been put into operation by major international companies within the pharmaceutical, medical devices, and orthopedic industries.
The process is validated and supported by existing regulations. As such, there are currently no limits to the incorporation of this technology, which represents the future of low-temperature sterilization. Presenting with ecological advantages and linked to reductions in production times and costs, HyPerPure® is a must-have for orthopedic device manufacturers.
De Lama laboratories are always open to run trials for customers specific products to verify complete sterilization of their devices as well as the compatibility of materials.

Click here to find out more about De Lama >>>>>












