While outsourcing may be commonplace within orthopedic device manufacturing, there are still opportunities to gain insights on best practices to optimize the process. While the industry has become more open to the opportunities and value these manufacturing partners offer, stumbling blocks still exist that infringe upon a successful relationship.
Companies and their design engineers need to remember to engage these manufacturing partners early in a product development process. If the contract manufacturer is already a trusted partner, they need to be considered an extension of the company, not an independent entity that is on a “need to know” basis. They exist to aid the OEM in their projects and guide them to a pathway toward success.
In the following interview, Ken Gredick, director of engineering at Triangle, offered a variety of factors orthopedic device OEMs need to keep in mind when beginning a project or just generally engaging with a contract manufacturer. The family-owned business based in Upper Saddle River, NJ, has been working with device manufacturers for more than 60 years.
Sean Fenske: What are the key criteria orthopedic device OEMs should keep in mind when selecting a contract manufacturing partner?
Ken Gredick: There are a number of criteria OEMs need to consider while selecting a true manufacturing partner.
First, the OEM needs to match up the type of device being outsourced to the supplier’s capabilities. Some buyers think custom manufactured components are commodities, and anyone can make their parts. This is a recipe for disaster. A few characteristics that need to be evaluated are the complexity of the design, the tightest tolerances on the print, geometric and dimensional tolerance, any non-verifiable processes, outside processes (plating, coating, etc.), just to name a few.
Of course, positive quality and delivery performance are also critical. But, a great supplier also has the ability to identify potential issues up front and offer solutions that work with the design intent. If this is a new device, it is best for the OEM to get the manufacturing partner involved early before the final design is complete. This gives the contract manufacturer the ability to give design feedback around the manufacturing and inspection procedures. Inspection is more important for the quality control department, but, depending on how the print is detailed, it can cause quality issues that turn into delivery issues. One simple example involves the measurement of a circle, which has a number of ways to be performed (as indicated in the illustration). If the print is not clearly defined and up to the quality engineer (or the inspector), there could be a measurement discrepancy, causing a reject and resulting in a loss of valuable time. Here at Triangle, we like to spend our time up front mitigating these issues so the customer has good product on time.
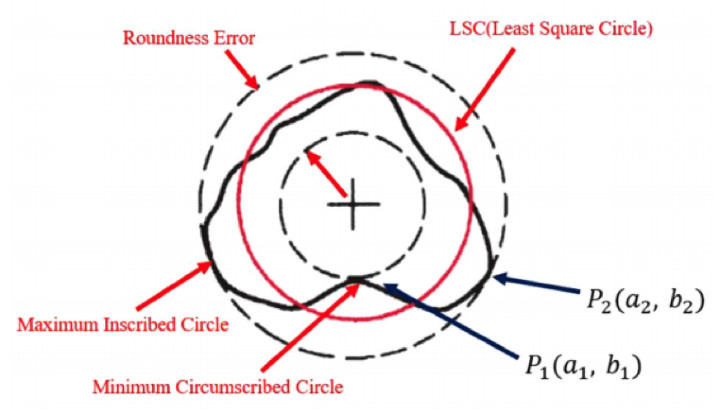
There can be a number of ways to inspect and measure something as simple as a circle.
Fenske: What are your best practices to ensure clear communication on a project between the contract manufacturer (i.e., yourself) and the orthopedic device OEM (i.e., your client)?
Gredick: Weekly status meetings to review completed tasks and open action items are very helpful. The high-level project plans will be discussed at these meetings along with any issues with the critical path.
Fenske: How often will an orthopedic device OEM come to you requesting a certain manufacturing process and you’re able to suggest less expensive and/or more reliable alternatives?
Gredick: This happens with design engineers who we have worked with in the past. They frequently run ideas by us as they are developing new parts or reach out to us when they are reviewing cost opportunities of current production.
Fenske: How often is the company open to your recommendations?
Gredick: Most companies are open to good feedback and implement them into the design.
Fenske: Can you please explain what Triangle’s Product Realization Process is and how it is tied to DFM (design for manufacturing) and DFI (design for inspection)?
Gredick: Triangle’s Product Realization Process is our process for developing a new product from inception to delivery. It promotes DFM and DFI, which optimize the prints for production. This process has proven to minimize change requests and production issues.
Fenske: Why is DFM and DFI important to consider early in the development process?
Gredick: The result of a proper DFM/DFI is savings. Some of the benefits are
- Reducing or eliminating expensive manufacturing techniques
- Reducing scrap because of optimized print callouts
- Streamlining inspection techniques
- Standardized tooling allows using off-the-shelf versus custom
- A manufacturable design, shorter lead times, and minimal changes
Once a process is developed and proved out, it is very costly to make a change in medical manufacturing. You may have to requalify your process, which may include performing capability studies, first article inspections, measurement systems analysis, etc. Doing the DFM/DFI up front streamlines the manufacturing process, avoiding Muda (i.e., eight wastes) and costly changes.
Fenske: How do you handle an orthopedic device OEM who comes to you with extremely tight tolerances for the entire component or implant that ultimately adds unnecessary costs to the project? Are you able to steer them in a better direction?
Gredick: Good question! In this situation, I like to understand the assembly and/or application to better understand why the tight tolerances are needed. This may bring to light a conversation where we can open up some tolerances. If not, we need to evaluate two paths: Can this be manufactured on a consistent basis (DFM)? Or, do we need to align inspection methodologies for repeatability, reproducibility, accuracy, and correlation (part of measurement system analysis, or MSA)?
The best way to handle these situations is to get the subject matter experts (SMEs) working collaboratively to come up with a solution that works for both companies. The SMEs typically consist of the design engineer, quality engineer, CMM programmer, CNC programmers, and/or the inspectors. Once everyone understands the full scope and everyone’s expertise, a detailed plan is put in place to create actions and deliverables. This team should be responsible throughout the full scope of the first manufacturing lot (at a minimum).
Fenske: Do you have any additional comments you’d like to share based on any of the topics we discussed or something you’d like to tell medical device designers?
Gredick: It is extremely important to engage the manufacturing partner on a project as early as you can. By doing this, the project’s timeline could shrink, and they would have a smaller risk of bad product manufactured.
Learn more about Triangle >>>>>












