01.28.15
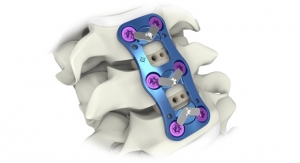
Carlsbad, Calif.-based Aurora Spine Corp. recently reported the first surgery in Nazareth, Israel, using the Eos minimally invasive transforaminal lumbar interbody fusion (TLIF) system featuring TiNano, the company’s titanium plasma spray coating. TLIF is a spinal fusion procedure that fuses the front and back section of the spine through a posterior approach. The procedure marked the first time a TiNano-coated interbody cage surgery was performed outside the United States.
The surgery was performed at the Nazareth Hospital E.M.M.S by Khaled Aslan, M.D.
"It was great to do the first Zip-Eos combination in Israel. The implants and instrumentation are very intuitive and easy to use, and they significantly shortened the operation time," said Aslan.
Eos is Aurora Spine's minimally invasive TLIF interbody fusion cage manufactured with PEEK-Optima and coated with TiNano.
The combination of the Eos TiNano coated cage along with the Zip Ultra MIS (minimally invasive surgery) fixation implant together comprise Aurora Spine's proprietary “Screwless Procedure.”
"Aurora is changing spine surgery," said Trent J. Northcutt, Aurora's president and CEO. "We designed the Zip MIS interspinous fusion system and TiNano coated interbody cages to improve spine patient outcomes, drive continued surgeon interest, and bolster our relevance as a key innovator in spine. Aurora Spine's minimally invasive Screwless Procedure is designed to provide unique benefits that deliver value to hospitals and patients around the world."
Aurora Spine is an early-stage spinal company focused on a series of screwless, minimally invasive, regenerative spinal implant technologies. The company recently obtained its fifth U.S. Food and Drug Administration clearance in the past 12 months for its Zip 51 invasive interspinous fixation implant for spinal fusion. The Zip 51 was developed as an alternative to pedicle screw fixation and designed for stabilization and load sharing during the TI-S1 thoracolumbar fusion procedures. The implant is also intended to be an adjunct to interbody fusion, specifically for the treatment of degenerative disc disease, spondylolisthesis, trauma, and/or tumor.
The surgery was performed at the Nazareth Hospital E.M.M.S by Khaled Aslan, M.D.
"It was great to do the first Zip-Eos combination in Israel. The implants and instrumentation are very intuitive and easy to use, and they significantly shortened the operation time," said Aslan.
Eos is Aurora Spine's minimally invasive TLIF interbody fusion cage manufactured with PEEK-Optima and coated with TiNano.
The combination of the Eos TiNano coated cage along with the Zip Ultra MIS (minimally invasive surgery) fixation implant together comprise Aurora Spine's proprietary “Screwless Procedure.”
"Aurora is changing spine surgery," said Trent J. Northcutt, Aurora's president and CEO. "We designed the Zip MIS interspinous fusion system and TiNano coated interbody cages to improve spine patient outcomes, drive continued surgeon interest, and bolster our relevance as a key innovator in spine. Aurora Spine's minimally invasive Screwless Procedure is designed to provide unique benefits that deliver value to hospitals and patients around the world."
Aurora Spine is an early-stage spinal company focused on a series of screwless, minimally invasive, regenerative spinal implant technologies. The company recently obtained its fifth U.S. Food and Drug Administration clearance in the past 12 months for its Zip 51 invasive interspinous fixation implant for spinal fusion. The Zip 51 was developed as an alternative to pedicle screw fixation and designed for stabilization and load sharing during the TI-S1 thoracolumbar fusion procedures. The implant is also intended to be an adjunct to interbody fusion, specifically for the treatment of degenerative disc disease, spondylolisthesis, trauma, and/or tumor.