05.27.15
San Clemente, Calif.-based VertiFlex Inc., which makes advanced minimally invasive interventions for spinal stenosis, has earned approval from the U.S. Food and Drug Administration (FDA) for its Superion Interspinous Spacer System, clearing the way for commercialization in the United States. The approval follows a February FDA advisory panel vote that Superion demonstrated safety, effectiveness, and a favorable risk benefit profile based on the results from a 470 patient, multi-center, prospective and randomized controlled investigational device exemption (IDE) trial.
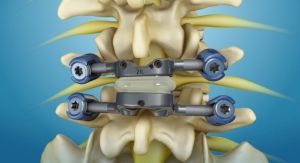
Superion is a minimally invasive, motion preserving interspinous spacer indicated for moderate lumbar spinal stenosis. The company reports that Superion demonstrated more than 80 percent clinical success in all major components of the composite primary endpoint of the IDE trial at 24 months, maintaining durability of effect through 36 months. Superion patients also reportedly exhibited similar leg pain improvement compared to published literature on open surgical decompression. The company also claims the device is cost-effective, comparable in cost to open surgical decompression.
VertiFlex anticipates the release of Superion in the United States soon.
“We are delighted with this approval based on compelling level one evidence developed in the Superion IDE trial,” said Earl R. Fender, president and CEO of VertiFlex. “Superion has proven to be a safe, effective and least invasive solution for the treatment of spinal stenosis, in a mostly Medicare-aged population. Countless numbers of patients in the United States will now benefit from this important technological advancement.”
“As an early adopter of interspinous spacers, they have provided tremendous improvement for my patients suffering from lumbar stenosis,” said Nick Shamie, M.D., professor and chief of orthopedic spine surgery, at the University of California – Los Angeles, and co-medical director for VertiFlex. “As a next-generation technology, Superion offers the potential for even greater clinical benefit, with the least invasive indirect decompression possible, and the ability for patients to avoid traditional open spine surgery.”
Superion is a minimally invasive, motion preserving interspinous spacer indicated for moderate lumbar spinal stenosis. The company reports that Superion demonstrated more than 80 percent clinical success in all major components of the composite primary endpoint of the IDE trial at 24 months, maintaining durability of effect through 36 months. Superion patients also reportedly exhibited similar leg pain improvement compared to published literature on open surgical decompression. The company also claims the device is cost-effective, comparable in cost to open surgical decompression.
VertiFlex anticipates the release of Superion in the United States soon.
“We are delighted with this approval based on compelling level one evidence developed in the Superion IDE trial,” said Earl R. Fender, president and CEO of VertiFlex. “Superion has proven to be a safe, effective and least invasive solution for the treatment of spinal stenosis, in a mostly Medicare-aged population. Countless numbers of patients in the United States will now benefit from this important technological advancement.”
“As an early adopter of interspinous spacers, they have provided tremendous improvement for my patients suffering from lumbar stenosis,” said Nick Shamie, M.D., professor and chief of orthopedic spine surgery, at the University of California – Los Angeles, and co-medical director for VertiFlex. “As a next-generation technology, Superion offers the potential for even greater clinical benefit, with the least invasive indirect decompression possible, and the ability for patients to avoid traditional open spine surgery.”