06.04.15
St. Paul, Minn.-based St. Jude Medical Inc. has earned CE mark approval for its Invisible Trial system, an app-based, wireless neuromodulation programming system that uses Apple iPod touch and iPad mini technology. Simultaneously with the CE mark, the system will be released in Europe. Like other Apple products, the new St. Jude Medical system relies on Bluetooth communication; the system is for trial spinal cord stimulation patients for the treatment of chronic pain prior to permanent implantation.
“I expect that the St. Jude Medical Invisible Trial system will significantly improve the trial experience for my patients,” said Stefan Schu, M.D., specialist for neurosurgery and senior physician for neuromodulation at the Sana Clinic in Duisburg, Germany. “The new system will be discreet, familiar and require no cables that can be uncomfortable or potentially cause the lead to dislodge. Perhaps the most important feature is the therapy itself, which will enable a unique burst stimulation mode that will expand the range of stimulation modes available in the trial phase and thereby potentially improve the trial success rate for my patients suffering from chronic pain.”
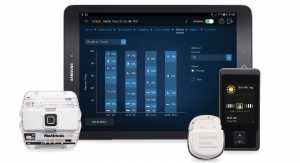
One of the key system features of the Invisible Trial system is the use of an external pulse generator (EPG) as the system’s power source. The EPG is small and uses Bluetooth communication to communicate between the patient’s iPod touch controller and the stimulation system. As a result, the system can now be worn under a patient’s clothing, often rendering the entire system “invisible,” which is hoped to provide a more comfortable trial experience. The goal is for patients to focus more on potential pain relief and therapeutic impact during their trial and less on the trial system itself.
The iPod touch controller offers patients a simple, familiar platform to adjust their therapy. An iPad mini tablet is used by the patient’s physician to set the programming parameters. The programmer also displays trial usage data from the EPG and allows the physician to print or email the data in PDF format. Bluetooth technology safely and securely communicates wirelessly between the EPG and patient and physician devices, eliminating the programming trial cable and thus increasing the patient’s comfort.
The new trial system has the capability to deliver both traditional and burst stimulation modes. Burst stimulation has been demonstrated to minimize paresthesia (pricking or tingling sensation) in most patients. Paresthesia can often fluctuate with posture and body position changes and negatively impact the trial experience. Being able to utilize the Burst stimulation mode in a trial setting expands the range of available stimulation modes for chronic pain sufferers in the trial phase and in addition offers the potential to trial Burst stimulation for patients who did not respond to traditional tonic stimulation previously.
“We’ve developed our new patient-centric Invisible Trial System as a response to physician and patient feedback,” said Eric S. Fain, M.D., group president of St. Jude Medical. “The system was designed to improve the comfort and usability of our system for patients evaluating spinal cord stimulation therapy to alleviate their chronic pain without focusing on potential barriers such as programming trial cables and systems with complex trial controls.”
“I expect that the St. Jude Medical Invisible Trial system will significantly improve the trial experience for my patients,” said Stefan Schu, M.D., specialist for neurosurgery and senior physician for neuromodulation at the Sana Clinic in Duisburg, Germany. “The new system will be discreet, familiar and require no cables that can be uncomfortable or potentially cause the lead to dislodge. Perhaps the most important feature is the therapy itself, which will enable a unique burst stimulation mode that will expand the range of stimulation modes available in the trial phase and thereby potentially improve the trial success rate for my patients suffering from chronic pain.”
One of the key system features of the Invisible Trial system is the use of an external pulse generator (EPG) as the system’s power source. The EPG is small and uses Bluetooth communication to communicate between the patient’s iPod touch controller and the stimulation system. As a result, the system can now be worn under a patient’s clothing, often rendering the entire system “invisible,” which is hoped to provide a more comfortable trial experience. The goal is for patients to focus more on potential pain relief and therapeutic impact during their trial and less on the trial system itself.
The iPod touch controller offers patients a simple, familiar platform to adjust their therapy. An iPad mini tablet is used by the patient’s physician to set the programming parameters. The programmer also displays trial usage data from the EPG and allows the physician to print or email the data in PDF format. Bluetooth technology safely and securely communicates wirelessly between the EPG and patient and physician devices, eliminating the programming trial cable and thus increasing the patient’s comfort.
The new trial system has the capability to deliver both traditional and burst stimulation modes. Burst stimulation has been demonstrated to minimize paresthesia (pricking or tingling sensation) in most patients. Paresthesia can often fluctuate with posture and body position changes and negatively impact the trial experience. Being able to utilize the Burst stimulation mode in a trial setting expands the range of available stimulation modes for chronic pain sufferers in the trial phase and in addition offers the potential to trial Burst stimulation for patients who did not respond to traditional tonic stimulation previously.
“We’ve developed our new patient-centric Invisible Trial System as a response to physician and patient feedback,” said Eric S. Fain, M.D., group president of St. Jude Medical. “The system was designed to improve the comfort and usability of our system for patients evaluating spinal cord stimulation therapy to alleviate their chronic pain without focusing on potential barriers such as programming trial cables and systems with complex trial controls.”