LDR Holding Corporation11.19.15
The North American Spine Society (NASS) has revised its formal coverage recommendation for cervical artificial disc replacement (CADR), LDR Holding Corporation executives announced.
The evidence-based coverage policy recommendation, recently released by NASS, now includes support for both one- and two-level CADR when clinically indicated (radiculopathy or myelopathy from disc degeneration following appropriate non-operative care). The Coverage Committee said the "rationale for coverage of CADR is based on the indications and results of many randomized controlled trials (RCTs) that have compared the procedure to what most would consider the gold standard surgical treatment, anterior cervical discectomy and fusion (ACDF)."
"NASS is a global multidisciplinary medical society that utilizes education, research and advocacy to foster the highest quality, ethical, value- and evidence-based spine care for patients, so its recommendation for both one and two-level cervical disc replacement as a treatment option for those patients who satisfy the clinical indications is a major milestone in the treatment of cervical disc degeneration," LDR President/CEO Christophe Lavigne said. "The revised NASS policy recommendation further validates the emergence of CADR as an alternative to ACDF in appropriate patients and should help to further expand coverage for the Mobi-C Cervical Disc for both one and two-level procedures in the United States."
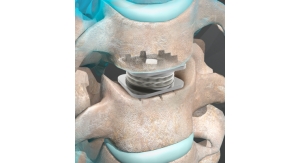
Mobi-C is a cobalt chromium alloy and polyethylene mobile-bearing prosthesis specifically designed as a bone-sparing, cervical intervertebral disc replacement for both one and two-level indications. All other cervical disc prostheses are FDA approved for one-level use only. In addition to the unique mobile-bearing feature, Mobi-C offers a simplified surgical technique.
The Mobi-C Cervical Disc Prosthesis is indicated in skeletally mature patients for reconstruction of the disc from C3-C7 following discectomy at one level or two contiguous levels for intractable radiculopathy (arm pain and/or neurological deficit) with or without neck pain, or myelopathy due to abnormality localized to the level of the disc space and at least one of the following conditions confirmed by radiographic imaging (CT, MRI, or X-rays): herniated nucleus pulposus, spondylosis (defined by the presence of osteophytes), and/or visible loss of disc height compared to adjacent levels. The Mobi-C Cervical Disc Prosthesis is implanted using an anterior approach. Patients should have failed at least six weeks of conservative treatment or demonstrated progressive signs or symptoms despite nonoperative treatment prior to implantation of the Mobi-C Cervical Disc Prosthesis.
With headquarter locations in Troyes, France, and Austin, Texas, LDR Holding Corporation designs and commercializes spinal surgery technologies. LDR's primary products are based on its exclusive Mobi non-fusion and VerteBRIDGE fusion technology platforms and are designed for applications in the cervical and lumbar spine. These technologies are designed to enable products that are less invasive, provide greater intra-operative flexibility, offer simplified surgical techniques and promote improved clinical outcomes for patients as compared to existing alternatives. In August 2013, LDR received approval from the U.S. Food and Drug Administration for the Mobi-C cervical disc replacement device.
The evidence-based coverage policy recommendation, recently released by NASS, now includes support for both one- and two-level CADR when clinically indicated (radiculopathy or myelopathy from disc degeneration following appropriate non-operative care). The Coverage Committee said the "rationale for coverage of CADR is based on the indications and results of many randomized controlled trials (RCTs) that have compared the procedure to what most would consider the gold standard surgical treatment, anterior cervical discectomy and fusion (ACDF)."
"NASS is a global multidisciplinary medical society that utilizes education, research and advocacy to foster the highest quality, ethical, value- and evidence-based spine care for patients, so its recommendation for both one and two-level cervical disc replacement as a treatment option for those patients who satisfy the clinical indications is a major milestone in the treatment of cervical disc degeneration," LDR President/CEO Christophe Lavigne said. "The revised NASS policy recommendation further validates the emergence of CADR as an alternative to ACDF in appropriate patients and should help to further expand coverage for the Mobi-C Cervical Disc for both one and two-level procedures in the United States."
Mobi-C is a cobalt chromium alloy and polyethylene mobile-bearing prosthesis specifically designed as a bone-sparing, cervical intervertebral disc replacement for both one and two-level indications. All other cervical disc prostheses are FDA approved for one-level use only. In addition to the unique mobile-bearing feature, Mobi-C offers a simplified surgical technique.
The Mobi-C Cervical Disc Prosthesis is indicated in skeletally mature patients for reconstruction of the disc from C3-C7 following discectomy at one level or two contiguous levels for intractable radiculopathy (arm pain and/or neurological deficit) with or without neck pain, or myelopathy due to abnormality localized to the level of the disc space and at least one of the following conditions confirmed by radiographic imaging (CT, MRI, or X-rays): herniated nucleus pulposus, spondylosis (defined by the presence of osteophytes), and/or visible loss of disc height compared to adjacent levels. The Mobi-C Cervical Disc Prosthesis is implanted using an anterior approach. Patients should have failed at least six weeks of conservative treatment or demonstrated progressive signs or symptoms despite nonoperative treatment prior to implantation of the Mobi-C Cervical Disc Prosthesis.
With headquarter locations in Troyes, France, and Austin, Texas, LDR Holding Corporation designs and commercializes spinal surgery technologies. LDR's primary products are based on its exclusive Mobi non-fusion and VerteBRIDGE fusion technology platforms and are designed for applications in the cervical and lumbar spine. These technologies are designed to enable products that are less invasive, provide greater intra-operative flexibility, offer simplified surgical techniques and promote improved clinical outcomes for patients as compared to existing alternatives. In August 2013, LDR received approval from the U.S. Food and Drug Administration for the Mobi-C cervical disc replacement device.