Active Implants LLC05.20.16
Active Implants LLC has completed the first meniscus replacement in its SUN (Safety Using NUsurface) clinical trial. The procedure was performed at Salt Lake Regional Medical Center in Salt Lake City, Utah, by Dr. Andrew Cooper.
Salt Lake Regional Medical Center is the only hospital in Utah and one of just 10 U.S. sites enrolling patients with persistent knee pain caused by injured or deteriorating meniscus cartilage in the SUN trial. The study is designed to assess the safety and effectiveness of the NUsurface Meniscus implant in restoring function similar to that of a natural, healthy meniscus.
The recipient of the implant was 55-year-old Uintah resident Kent Carlson, who has experienced meniscus-related problems since he was a child as a result of skiing injuries. He underwent a meniscectomy and has been getting cortisone shots, but the pain has persisted to the point where sitting for long periods or bearing weight are uncomfortable. Even low-impact exercises like swimming and cycling can cause his knee to swell.
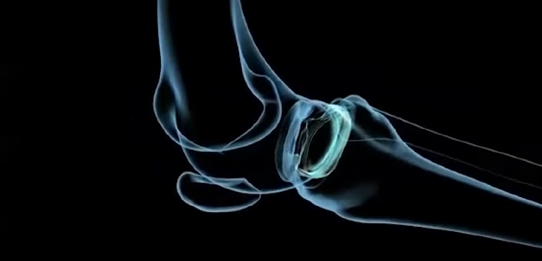
The meniscus is a tissue pad between the thigh and shin bones. Once damaged, the meniscus has a very limited ability to heal. More than 1 million partial meniscectomies to remove or repair a torn meniscus are performed in the United States annually, about the same as the total number of hip and knee replacement surgeries combined. However, many patients still experience persistent knee pain following meniscus surgery.
“There aren’t many options for patients like Kent, who experienced persistent knee pain following meniscus surgery,” said Cooper, orthopedic surgeon at Salt Lake Regional Medical Center. “Damage to the meniscus can lead to arthritis and the need for knee replacement surgery. We hope the NUsurface implant alleviates pain in these patients, as well as helps them delay or avoid knee replacement surgery.”
The NUsurface Meniscus Implant is inserted into the knee joint through a small incision and patients typically can go home soon after the operation. After surgery, they undergo a six-week rehabilitation program. The NUsurface Meniscus Implant has been used in Europe under CE Mark since 2008 and Israel since 2011.
Watch the video below to learn more about the NUsurface implant:
An avid outdoorsman, Carlson is looking forward to getting back into the wild once he recovers from the NUsurface surgery. “I love to hunt, fish and hike,” Carlson said. “I can’t wait to put on a pack and get back out there.”
The SUN study will enroll approximately 120 patients as part of regulatory process to gain approval from U.S. Food and Drug Administration to sell the device in the United States. All patients who meet study requirements and agree to enter the trial are offered the NUsurface Meniscus Implant as treatment. Treatment with NUsurface in the SUN trial is eligible for coverage by Medicare and some private insurance companies. To be eligible for the study, participants must be between the ages of 30 and 75 and have pain after medial meniscus surgery that was performed at least six months ago.
The NUsurface Meniscus implant is made from medical grade plastic and, as a result of its materials, composite structure and design, does not require fixation to bone or soft tissues. The NUsurface implant mimics the function of the natural meniscus and redistributes loads transmitted across the knee joint. Clinical trials are underway in the United States, Europe and Israel to verify the safety and effectiveness of the NUsurface Meniscus Implant.
Active Implants LLC develops orthopedic implant solutions that complement the natural biomechanics of the musculoskeletal system, allowing patients to maintain or return to an active lifestyle. Active Implants is privately held with headquarters in Memphis, Tenn. European offices are in Driebergen, The Netherlands, with R&D facilities in Netanya, Israel.
Salt Lake Regional Medical Center is the only hospital in Utah and one of just 10 U.S. sites enrolling patients with persistent knee pain caused by injured or deteriorating meniscus cartilage in the SUN trial. The study is designed to assess the safety and effectiveness of the NUsurface Meniscus implant in restoring function similar to that of a natural, healthy meniscus.
The recipient of the implant was 55-year-old Uintah resident Kent Carlson, who has experienced meniscus-related problems since he was a child as a result of skiing injuries. He underwent a meniscectomy and has been getting cortisone shots, but the pain has persisted to the point where sitting for long periods or bearing weight are uncomfortable. Even low-impact exercises like swimming and cycling can cause his knee to swell.
The meniscus is a tissue pad between the thigh and shin bones. Once damaged, the meniscus has a very limited ability to heal. More than 1 million partial meniscectomies to remove or repair a torn meniscus are performed in the United States annually, about the same as the total number of hip and knee replacement surgeries combined. However, many patients still experience persistent knee pain following meniscus surgery.
“There aren’t many options for patients like Kent, who experienced persistent knee pain following meniscus surgery,” said Cooper, orthopedic surgeon at Salt Lake Regional Medical Center. “Damage to the meniscus can lead to arthritis and the need for knee replacement surgery. We hope the NUsurface implant alleviates pain in these patients, as well as helps them delay or avoid knee replacement surgery.”
The NUsurface Meniscus Implant is inserted into the knee joint through a small incision and patients typically can go home soon after the operation. After surgery, they undergo a six-week rehabilitation program. The NUsurface Meniscus Implant has been used in Europe under CE Mark since 2008 and Israel since 2011.
Watch the video below to learn more about the NUsurface implant:
An avid outdoorsman, Carlson is looking forward to getting back into the wild once he recovers from the NUsurface surgery. “I love to hunt, fish and hike,” Carlson said. “I can’t wait to put on a pack and get back out there.”
The SUN study will enroll approximately 120 patients as part of regulatory process to gain approval from U.S. Food and Drug Administration to sell the device in the United States. All patients who meet study requirements and agree to enter the trial are offered the NUsurface Meniscus Implant as treatment. Treatment with NUsurface in the SUN trial is eligible for coverage by Medicare and some private insurance companies. To be eligible for the study, participants must be between the ages of 30 and 75 and have pain after medial meniscus surgery that was performed at least six months ago.
The NUsurface Meniscus implant is made from medical grade plastic and, as a result of its materials, composite structure and design, does not require fixation to bone or soft tissues. The NUsurface implant mimics the function of the natural meniscus and redistributes loads transmitted across the knee joint. Clinical trials are underway in the United States, Europe and Israel to verify the safety and effectiveness of the NUsurface Meniscus Implant.
Active Implants LLC develops orthopedic implant solutions that complement the natural biomechanics of the musculoskeletal system, allowing patients to maintain or return to an active lifestyle. Active Implants is privately held with headquarters in Memphis, Tenn. European offices are in Driebergen, The Netherlands, with R&D facilities in Netanya, Israel.