ASTM08.12.16
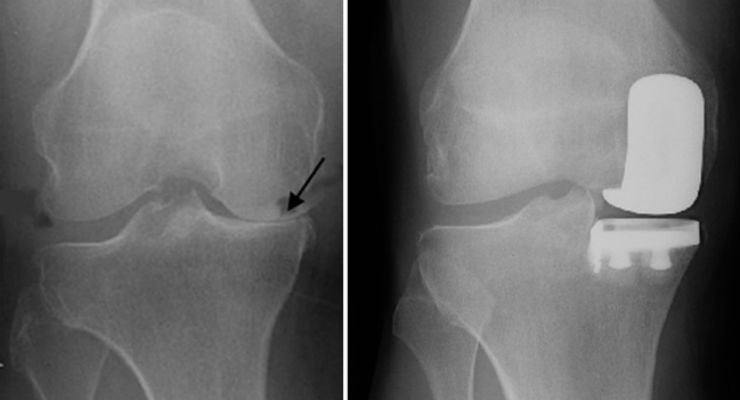
A proposed ASTM International test method will educate implant designers on the material properties and capabilities of implants, specifically those used in unicondylar knee joint replacements. The proposed standard (WK45235, Practice for Cyclic Fatigue Testing of Metal Tibial Tray Components of Unicondylar Knee Joint Replacements) is being developed by ASTM Committee F04 on Medical and Surgical Materials and Devices.
ASTM member Gokce Yildirim, of Stryker Orthopaedics, said “The standard is primarily for implant designers who would like to use the test method for physical and FEA applications to gain insight into the implant capabilities.”
ASTM welcomes participation in the development of its standards. Become a member atwww.astm.org/JOIN.
ASTM member Gokce Yildirim, of Stryker Orthopaedics, said “The standard is primarily for implant designers who would like to use the test method for physical and FEA applications to gain insight into the implant capabilities.”
ASTM welcomes participation in the development of its standards. Become a member atwww.astm.org/JOIN.