Business Wire08.15.16
Titan Spine, a medical device surface technology company focused on developing spinal interbody fusion implants, today announced that it has expanded its distribution agreement with strategic partner MBA to provide Titan’s line of Endoskeleton titanium implants to practicing spine surgeons in the European Union to include Italy.
Under the agreement, initially announced in September 2015, Titan Spine provides its spinal interbody fusion devices featuring its proprietary surface technology to MBA for distribution in seven EU countries: Spain, Portugal, Andorra, Luxembourg, Belgium, the United Kingdom (UK), and now Italy. MBA is a prominent international medical device marketing-and-distribution organization that specializes in the sale of products used for orthopedic and neurosurgery treatments.
Marcus Klarl, Vice President of Sales, Europe for Titan Spine, commented, “Our initial distribution agreement with MBA has proven largely successful over the past year due to accelerating sales growth. Increased surgeon adoption within the EU interbody device market has demonstrated the growing desire for surface enhanced interbody fusion devices that stimulate healing at the cellular level where it is most critical for improved patient outcomes. We look forward to providing these benefits to spine surgeons and patients in Italy—an interbody fusion market that is projected to surpass $25 million next year.”
“Titan Spine is more than a supplier for us; they are a partner,” commented Carlos Marina, CEO for MBA. “Since the beginning of our commercial relationship with Titan, we have worked together seamlessly. In fact, on introducing Titan Spine into the Italian market they have now become the only supplier that MBA works with in all countries where we operate: Spain, Andorra, Portugal, Belgium, UK, Luxembourg, and Italy.”
Paul Griffin, International Business Manager of MBA, added, “We are delighted with the level of success achieved to date with distribution of Titan Spine spinal implants in the countries where MBA has initially introduced the unique portfolio. Now, with the addition of distribution in Italy, MBA and Titan Spine are together strongly positioned to increase Italian market share with a comprehensive, spinal product offering.”
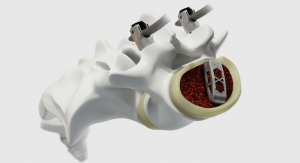
Titan Spine offers a full line of Endoskeleton devices that feature Titan Spine’s proprietary implant surface technology, consisting of a unique combination of roughened topographies at the macro, micro, and nano levels (MMN). This unique combination of surface topographies is designed to create an optimal host-bone response and actively participate in the fusion process by promoting the upregulation of osteogenic and angiogenic factors necessary for bone growth, encouraging natural production of bone morphogenetic proteins (BMPs), downregulating inflammatory factors, and creating the potential for a faster and more robust fusion.1,2,3,4 All Endoskeleton devices are covered by the company’s risk share warranty.
References
1Olivares-Navarrete, R., Hyzy, S.L., Slosar, P.J., Schneider, J.M., Schwartz, Z., and Boyan, B.D. (2015). Implant materials generate different peri-implant inflammatory factors: PEEK promotes fibrosis and micro-textured titanium promotes osteogenic factors. Spine, Volume 40, Issue 6, 399–404.
2Olivares-Navarrete, R., Gittens, R.A., Schneider, J.M., Hyzy, S.L., Haithcock, D.A., Ullrich, P.F., Schwartz, Z., Boyan, B.D. (2012). Osteoblasts exhibit a more differentiated phenotype and increased bone morphogenetic production on titanium alloy substrates than poly-ether-ether-ketone. The Spine Journal, 12, 265-272.
3Olivares-Navarrete, R., Hyzy, S.L., Gittens, R.A., Schneider, J.M., Haithcock, D.A., Ullrich, P.F., Slosar, P. J., Schwartz, Z., Boyan, B.D. (2013). Rough titanium alloys regulate osteoblast production of angiogenic factors. The Spine Journal, 13, 1563-1570.
4Olivares-Navarrete, R., Hyzy S.L., Gittens, R.A., Berg, M.E., Schneider, J.M., Hotchkiss, K., Schwartz, Z., Boyan, B. D. Osteoblast lineage cells can discriminate microscale topographic features on titanium-aluminum-vanadium surfaces. Ann Biomed Eng. 2014 Dec; 42 (12): 2551-61.
Under the agreement, initially announced in September 2015, Titan Spine provides its spinal interbody fusion devices featuring its proprietary surface technology to MBA for distribution in seven EU countries: Spain, Portugal, Andorra, Luxembourg, Belgium, the United Kingdom (UK), and now Italy. MBA is a prominent international medical device marketing-and-distribution organization that specializes in the sale of products used for orthopedic and neurosurgery treatments.
Marcus Klarl, Vice President of Sales, Europe for Titan Spine, commented, “Our initial distribution agreement with MBA has proven largely successful over the past year due to accelerating sales growth. Increased surgeon adoption within the EU interbody device market has demonstrated the growing desire for surface enhanced interbody fusion devices that stimulate healing at the cellular level where it is most critical for improved patient outcomes. We look forward to providing these benefits to spine surgeons and patients in Italy—an interbody fusion market that is projected to surpass $25 million next year.”
“Titan Spine is more than a supplier for us; they are a partner,” commented Carlos Marina, CEO for MBA. “Since the beginning of our commercial relationship with Titan, we have worked together seamlessly. In fact, on introducing Titan Spine into the Italian market they have now become the only supplier that MBA works with in all countries where we operate: Spain, Andorra, Portugal, Belgium, UK, Luxembourg, and Italy.”
Paul Griffin, International Business Manager of MBA, added, “We are delighted with the level of success achieved to date with distribution of Titan Spine spinal implants in the countries where MBA has initially introduced the unique portfolio. Now, with the addition of distribution in Italy, MBA and Titan Spine are together strongly positioned to increase Italian market share with a comprehensive, spinal product offering.”
Titan Spine offers a full line of Endoskeleton devices that feature Titan Spine’s proprietary implant surface technology, consisting of a unique combination of roughened topographies at the macro, micro, and nano levels (MMN). This unique combination of surface topographies is designed to create an optimal host-bone response and actively participate in the fusion process by promoting the upregulation of osteogenic and angiogenic factors necessary for bone growth, encouraging natural production of bone morphogenetic proteins (BMPs), downregulating inflammatory factors, and creating the potential for a faster and more robust fusion.1,2,3,4 All Endoskeleton devices are covered by the company’s risk share warranty.
References
1Olivares-Navarrete, R., Hyzy, S.L., Slosar, P.J., Schneider, J.M., Schwartz, Z., and Boyan, B.D. (2015). Implant materials generate different peri-implant inflammatory factors: PEEK promotes fibrosis and micro-textured titanium promotes osteogenic factors. Spine, Volume 40, Issue 6, 399–404.
2Olivares-Navarrete, R., Gittens, R.A., Schneider, J.M., Hyzy, S.L., Haithcock, D.A., Ullrich, P.F., Schwartz, Z., Boyan, B.D. (2012). Osteoblasts exhibit a more differentiated phenotype and increased bone morphogenetic production on titanium alloy substrates than poly-ether-ether-ketone. The Spine Journal, 12, 265-272.
3Olivares-Navarrete, R., Hyzy, S.L., Gittens, R.A., Schneider, J.M., Haithcock, D.A., Ullrich, P.F., Slosar, P. J., Schwartz, Z., Boyan, B.D. (2013). Rough titanium alloys regulate osteoblast production of angiogenic factors. The Spine Journal, 13, 1563-1570.
4Olivares-Navarrete, R., Hyzy S.L., Gittens, R.A., Berg, M.E., Schneider, J.M., Hotchkiss, K., Schwartz, Z., Boyan, B. D. Osteoblast lineage cells can discriminate microscale topographic features on titanium-aluminum-vanadium surfaces. Ann Biomed Eng. 2014 Dec; 42 (12): 2551-61.