Marketwired09.15.16
Medovex Corp. has entered into an international distribution agreement with M.Fast Technologies Ltd., a supplier of spine surgery products. The agreement covers the distribution of the DenerveX System throughout Israel.
“M.Fast Technologies Ltd. serves as a perfect call point fit for our mission to provide a successful distribution, sales and marketing foundation for our entry of the DenerveX System in the Middle East,” said Dennis Moon, Medovex executive vice president.
Medovex already has distribution agreements for the DenerveX System in Germany, Spain, the United Kingdom, Denmark, Norway, Sweden, Finland in Europe and Asia Pacific, as well as in select other countries around the world.
“With M.Fast Technologies as a specialized distributor offering complex spine products throughout Israel, we are closing a gap while showing a professional presence in a major market,” added Manfred Sablowski, senior vice president of sales and marketing. “Today’s news importantly enables Medovex to expand the use of the DenerveX System into the Middle East, where M.Fast Technologies is a well established successful distributor. We expect the agreement will help in our efforts to increase worldwide adoption of the DenerveX System. Our goal remains to continuously improve our international market presence by expanding our network of strong distributors with knowledgeable sales and support personnel, and in close proximity to our customers.”
Medovex’s patented DenerveX System, currently in final development and not yet commercially available, is designed to provide longer lasting relief of pain associated with the facet joint. Lower back pain is the second most common cause of disability in the United States for adults. Studies indicate that 10 percent of the U.S. adult population suffers from lower back pain and that 31 percent of lower back pain is attributed to facet joint pain.
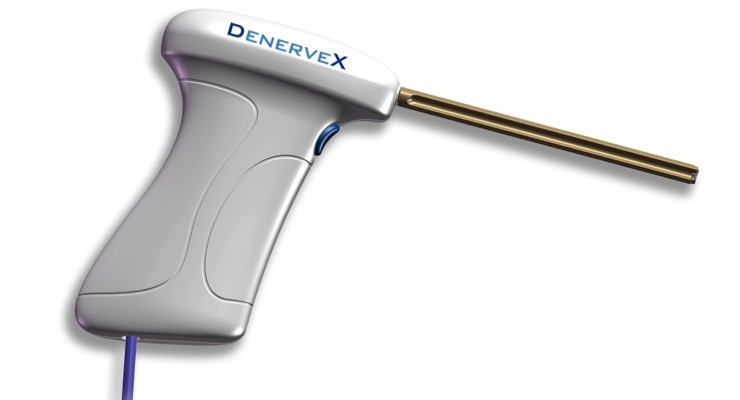
The DenerveX System consists of the DenerveX device kit containing a single use device, and the DenerveX Pro-40 Power Generator. The DenerveX System is designed to provide a minimally invasive treatment option which combines two actions into one device.
DenerveX is not yet CE marked or U.S. Food and Drug Administration-cleared and is not yet commercially available.
Medovex was formed to acquire and develop a diversified portfolio of medical technology products. Criteria for selection include those products with potential for significant improvement in the quality of patient care combined with cost effectiveness. The Atlanta, Ga.-based company’s first pipeline product, the DenerveX device, is intended to provide long lasting relief from pain associated with facet joint syndrome at significantly less cost than currently available options.
M.Fast Technologies is a supplier of spine surgery products.
“M.Fast Technologies Ltd. serves as a perfect call point fit for our mission to provide a successful distribution, sales and marketing foundation for our entry of the DenerveX System in the Middle East,” said Dennis Moon, Medovex executive vice president.
Medovex already has distribution agreements for the DenerveX System in Germany, Spain, the United Kingdom, Denmark, Norway, Sweden, Finland in Europe and Asia Pacific, as well as in select other countries around the world.
“With M.Fast Technologies as a specialized distributor offering complex spine products throughout Israel, we are closing a gap while showing a professional presence in a major market,” added Manfred Sablowski, senior vice president of sales and marketing. “Today’s news importantly enables Medovex to expand the use of the DenerveX System into the Middle East, where M.Fast Technologies is a well established successful distributor. We expect the agreement will help in our efforts to increase worldwide adoption of the DenerveX System. Our goal remains to continuously improve our international market presence by expanding our network of strong distributors with knowledgeable sales and support personnel, and in close proximity to our customers.”
Medovex’s patented DenerveX System, currently in final development and not yet commercially available, is designed to provide longer lasting relief of pain associated with the facet joint. Lower back pain is the second most common cause of disability in the United States for adults. Studies indicate that 10 percent of the U.S. adult population suffers from lower back pain and that 31 percent of lower back pain is attributed to facet joint pain.
The DenerveX System consists of the DenerveX device kit containing a single use device, and the DenerveX Pro-40 Power Generator. The DenerveX System is designed to provide a minimally invasive treatment option which combines two actions into one device.
DenerveX is not yet CE marked or U.S. Food and Drug Administration-cleared and is not yet commercially available.
Medovex was formed to acquire and develop a diversified portfolio of medical technology products. Criteria for selection include those products with potential for significant improvement in the quality of patient care combined with cost effectiveness. The Atlanta, Ga.-based company’s first pipeline product, the DenerveX device, is intended to provide long lasting relief from pain associated with facet joint syndrome at significantly less cost than currently available options.
M.Fast Technologies is a supplier of spine surgery products.