Business Wire01.16.17
SpineGuard, a developer and marketer of disposable surgical devices, has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its new DSG (Dynamic Surgical Guidance) integration module to be used in combination with Zavation’s spinal fusion system to make its pedicle screws “smart.”
“This FDA clearance will allow U.S. spine surgeons to insert pedicle screws ‘with a DSG enabled device’ not only in just one step but also with confidence, hence further securing and streamlining the most commonly performed instrumented spinal procedure, fusion. We are thrilled to immediately begin supplying Zavation, our U.S. partner, with our DSG integration module and look forward to working with our combined networks for the introduction of the first DSG-enabled pedicle screw in the U.S. market,” said Pierre Jérôme, CEO and co-founder of SpineGuard.
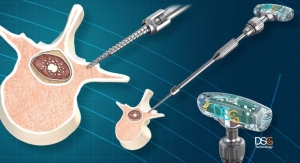
A DSG-enabled screw is the unique combination of a bipolar sensor and a pedicle screw in just one device. The technology offers surgeons real-time guidance and the ability to insert the screw directly into a vertebra without drilling a pilot hole. In minimally invasive surgery, it also obviates the need for a k-wire.
The DSG sensor differentiates various tissue types (cancellous bone, cortical bone, blood and soft tissues) based on the analysis of the local electrical conductivity. Real-time feedback informs the surgeon of changes in tissue type by an audio signal varying in pitch and cadence. This in turn alerts the surgeon of potential breaches during pedicle screw placement and allows for screw redirection.
The outcome is a single-step pedicle screw insertion with a high degree of accuracy, reduced radiation exposure and streamlined surgical steps resulting in potential time and cost savings.
Another DSG-enabled SmartScrew co-developed with Neuro France Implants (La ville-aux-Clercs, France) is currently in alpha launch in Europe, with seven surgeons having started to use the system.
“DSG-enabled devices offer a new paradigm to pedicle screw manufacturers who wish to differentiate their products from the rank-and-file screws in the market today. We are extremely pleased that our first DSG-partner in the U.S. is Zavation, and we look forward to extending this technology platform to other players in the industry," said Stéphane Bette, co-founder, chief technology officer and U.S. general manager of SpineGuard.
Co-founded in 2009 in France and the United States by Jérôme and Bette, SpineGuard aims to make spine surgery safer by bringing real-time digital technology into the operating room. Its primary objective is to establish its proprietary DSG technology as the global standard of surgical care, starting with safer screw placement in spine surgery and then in other surgeries. PediGuard, the first device designed using DSG, was co-invented by Maurice Bourlion, Ph.D., Ciaran Bolger, M.D., Ph.D., and Alain Vanquaethem, Biomedical Engineer. It is the world’s first and only handheld device capable of alerting surgeons to potential pedicular or vertebral breaches, according to the company. More than 50,000 surgical procedures have been performed worldwide with DSG enabled devices. Numerous studies published in peer-reviewed medical and scientific journals have demonstrated the multiple benefits that PediGuard delivers to patients, surgical staff and hospitals. SpineGuard is expanding the scope of its DSG platform through strategic partnerships with innovative medical device companies and the development of smart instruments and implants. SpineGuard has offices in San Francisco, Calif., and Paris, France.
“This FDA clearance will allow U.S. spine surgeons to insert pedicle screws ‘with a DSG enabled device’ not only in just one step but also with confidence, hence further securing and streamlining the most commonly performed instrumented spinal procedure, fusion. We are thrilled to immediately begin supplying Zavation, our U.S. partner, with our DSG integration module and look forward to working with our combined networks for the introduction of the first DSG-enabled pedicle screw in the U.S. market,” said Pierre Jérôme, CEO and co-founder of SpineGuard.
A DSG-enabled screw is the unique combination of a bipolar sensor and a pedicle screw in just one device. The technology offers surgeons real-time guidance and the ability to insert the screw directly into a vertebra without drilling a pilot hole. In minimally invasive surgery, it also obviates the need for a k-wire.
The DSG sensor differentiates various tissue types (cancellous bone, cortical bone, blood and soft tissues) based on the analysis of the local electrical conductivity. Real-time feedback informs the surgeon of changes in tissue type by an audio signal varying in pitch and cadence. This in turn alerts the surgeon of potential breaches during pedicle screw placement and allows for screw redirection.
The outcome is a single-step pedicle screw insertion with a high degree of accuracy, reduced radiation exposure and streamlined surgical steps resulting in potential time and cost savings.
Another DSG-enabled SmartScrew co-developed with Neuro France Implants (La ville-aux-Clercs, France) is currently in alpha launch in Europe, with seven surgeons having started to use the system.
“DSG-enabled devices offer a new paradigm to pedicle screw manufacturers who wish to differentiate their products from the rank-and-file screws in the market today. We are extremely pleased that our first DSG-partner in the U.S. is Zavation, and we look forward to extending this technology platform to other players in the industry," said Stéphane Bette, co-founder, chief technology officer and U.S. general manager of SpineGuard.
Co-founded in 2009 in France and the United States by Jérôme and Bette, SpineGuard aims to make spine surgery safer by bringing real-time digital technology into the operating room. Its primary objective is to establish its proprietary DSG technology as the global standard of surgical care, starting with safer screw placement in spine surgery and then in other surgeries. PediGuard, the first device designed using DSG, was co-invented by Maurice Bourlion, Ph.D., Ciaran Bolger, M.D., Ph.D., and Alain Vanquaethem, Biomedical Engineer. It is the world’s first and only handheld device capable of alerting surgeons to potential pedicular or vertebral breaches, according to the company. More than 50,000 surgical procedures have been performed worldwide with DSG enabled devices. Numerous studies published in peer-reviewed medical and scientific journals have demonstrated the multiple benefits that PediGuard delivers to patients, surgical staff and hospitals. SpineGuard is expanding the scope of its DSG platform through strategic partnerships with innovative medical device companies and the development of smart instruments and implants. SpineGuard has offices in San Francisco, Calif., and Paris, France.