PR Web01.27.17
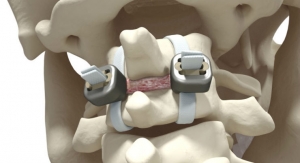
VTI, a minimally invasive surgical medical device company focused on lumber spine solutions, has been granted a new patent from the U.S. Patent and Trademark Office. This patent relates to the company’s modular, in vivo assembly technology and its motion preservation, InterCushion pipeline product. VTI now has more than 25 patents and patent pending assets in its global intellectual property portfolio.
“The grant of this patent expands and lengthens VTI’s protection of our in vivo expansion technology for minimally invasive lumbar spine solutions. VTI is a leader in lumbar spine focused motion preservation/disc nucleus replacement technology. This patent exemplifies our continued commitment to protect and broaden our intellectual property portfolio in this space. We are encouraged by the clinical results we are seeing from clinical trials using VTI’s modular technology in our InterCushion product line,” said Matthew R Kyle, president and CEO of VTI.
VTI’s InterCushion lumbar spine motion preservation product has been successful in reducing pain, preserving disc, and halting modic changes in clinical trials in Canada. Currently, several patients are at or near five years’ post implantation with continued clinically successful outcomes.
Dr. Philip de Muelenaere said, “The potential value of an intervertebral nucleus replacement is linked to decreased future deterioration of the spinal motion segment. By maintaining disc height, the motion is maintained, stability is regained and long term collapse prevented. This protects the patency of the foramina, and decreases facet arthrosis which, apart from being a pain generator, will compromise the exiting nerve root. The five-year follow up magnetic resonance imaging study proves that the prosthesis had maintained its position, maintained the disc height and kept the patients pain free.”
VTI – Vertebral Technologies Inc. is a privately held company based in Minneapolis, Minn. VTI is dedicated to the design, development, manufacturing and marketing of medical devices to address painful conditions of the spine through less-invasive surgical approaches. VTI’s products utilize its modular-assembly technology to deliver solutions optimized for both surgeons and their patients.
“The grant of this patent expands and lengthens VTI’s protection of our in vivo expansion technology for minimally invasive lumbar spine solutions. VTI is a leader in lumbar spine focused motion preservation/disc nucleus replacement technology. This patent exemplifies our continued commitment to protect and broaden our intellectual property portfolio in this space. We are encouraged by the clinical results we are seeing from clinical trials using VTI’s modular technology in our InterCushion product line,” said Matthew R Kyle, president and CEO of VTI.
VTI’s InterCushion lumbar spine motion preservation product has been successful in reducing pain, preserving disc, and halting modic changes in clinical trials in Canada. Currently, several patients are at or near five years’ post implantation with continued clinically successful outcomes.
Dr. Philip de Muelenaere said, “The potential value of an intervertebral nucleus replacement is linked to decreased future deterioration of the spinal motion segment. By maintaining disc height, the motion is maintained, stability is regained and long term collapse prevented. This protects the patency of the foramina, and decreases facet arthrosis which, apart from being a pain generator, will compromise the exiting nerve root. The five-year follow up magnetic resonance imaging study proves that the prosthesis had maintained its position, maintained the disc height and kept the patients pain free.”
VTI – Vertebral Technologies Inc. is a privately held company based in Minneapolis, Minn. VTI is dedicated to the design, development, manufacturing and marketing of medical devices to address painful conditions of the spine through less-invasive surgical approaches. VTI’s products utilize its modular-assembly technology to deliver solutions optimized for both surgeons and their patients.