Business Wire03.13.17
Cartiva Inc., a developer of products for treating cartilage damage and osteoarthritis, today announced publication of five-year data for Cartiva Synthetic Cartilage Implant (SCI) in the March issue of Foot and Ankle International, the peer-reviewed journal of the American Orthopaedic Foot and Ankle Society. The publication, titled “Midterm Outcomes of Polyvinyl Alcohol Hydrogel Hemiarthroplasty of the First Metatarsophalangeal Joint in Advanced Hallux Rigidus,” presented results from 27 patients at three Canadian sites that participated in the Company’s 236-patient prospective, randomized multi-center, pivotal, clinical trial.
Key findings of the paper include:
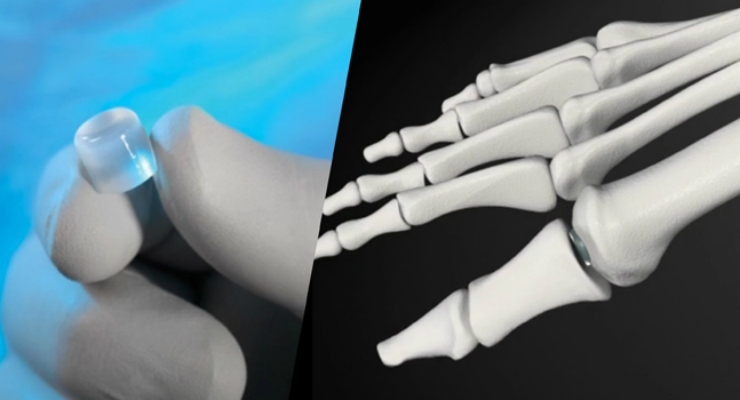
Cartiva SCI is intended to treat painful arthritis at the base of the great toe—known as hallux rigidus—the most common arthritic condition in the foot. The current standard of care fuses the arthritic joint bones with screws and plates. While effective at eliminating pain, it permanently prevents movement of the joint.
Cartiva SCI is a biocompatible polymer implant designed to have physical properties similar to articular cartilage. Damaged cartilage is replaced with a small Cartiva SCI implant that provides a compressible, durable, low-friction surface.
Cartiva SCI received Food and Drug Administration (FDA) premarket approval in July 2016, the first and only synthetic cartilage device approved by FDA.
The paper’s lead author, Timothy R. Daniels, M.D., FRCSC, Full Professor, University of Toronto and Head, Division of Orthopaedic Surgery, St. Michael's Hospital, said “These clinical results are consistent with what I have seen in my own practice. Pain and functional outcomes improved significantly, and 96% of the patients said they would choose to undergo the procedure again.”
“Cartiva SCI has been used commercially outside the United States for nearly l5 years, and publication of its long-term durability in this prestigious, peer-reviewed journal is another key milestone,” said Tim Patrick, president and CEO, Cartiva, Inc. “We will continue adding to the unparalleled, Level 1 clinical evidence of Cartiva SCI as a motion-preserving alternative to joint fusion.”
Key findings of the paper include:
- Patients experienced durable pain relief, achieving a 91% mean reduction in pain at 5 years.
- Functional improvement was sustained with patients demonstrating a 128% mean improvement in sporting activities.
- Patients experienced a 42% improvement in range of motion from baseline.
- Patients exhibited a high rate of satisfaction with the treatment.
Cartiva SCI is intended to treat painful arthritis at the base of the great toe—known as hallux rigidus—the most common arthritic condition in the foot. The current standard of care fuses the arthritic joint bones with screws and plates. While effective at eliminating pain, it permanently prevents movement of the joint.
Cartiva SCI is a biocompatible polymer implant designed to have physical properties similar to articular cartilage. Damaged cartilage is replaced with a small Cartiva SCI implant that provides a compressible, durable, low-friction surface.
Cartiva SCI received Food and Drug Administration (FDA) premarket approval in July 2016, the first and only synthetic cartilage device approved by FDA.
The paper’s lead author, Timothy R. Daniels, M.D., FRCSC, Full Professor, University of Toronto and Head, Division of Orthopaedic Surgery, St. Michael's Hospital, said “These clinical results are consistent with what I have seen in my own practice. Pain and functional outcomes improved significantly, and 96% of the patients said they would choose to undergo the procedure again.”
“Cartiva SCI has been used commercially outside the United States for nearly l5 years, and publication of its long-term durability in this prestigious, peer-reviewed journal is another key milestone,” said Tim Patrick, president and CEO, Cartiva, Inc. “We will continue adding to the unparalleled, Level 1 clinical evidence of Cartiva SCI as a motion-preserving alternative to joint fusion.”