Business Wire03.13.17
Spineology Inc. is excited to announce expanded FDA clearance of the Elite Expandable Interbody Fusion System. Included in the clearance is the addition of a narrower, 10mm, version of the device and an indication to utilize the devices with allograft bone.
“The broader size offering and the allograft bone indication allow the surgeon to pair Elite with Incite Cortical Fibers,” said John Booth, CEO of Spineology. “Incite’s excellent handling properties and ability to expand in-situ with the Elite Implant are added benefits not found in competitive systems.”
“The Elite Expandable Interbody device is an ideal choice when treating patients with a minimally invasive approach,” said Dr. Mark Grubb of Cleveland, Ohio. “The minimal initial starting height and a 10mm wide footprint reduces neural retraction requirements. Additionally, having the Incite bone graft that expands with the implant helps ensure good graft apposition.”
Dr. Dwight Tyndall of Munster, Ind., uses Elite with the Spineology ACT MediaLIF posterior mid-line approach. “The multiple width, length, and lordosis options make Elite an excellent choice for the mid-line approach and bilateral implant placement,” said Dr. Tyndall.
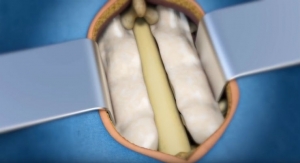
The Elite Expandable Interbody Fusion implant is inserted into the disc space at a contracted minimum height and is expanded once in position to restore anatomic disc height, provide anterior column support, and potentially facilitate indirect decompression. Elite offers the surgeon the ability to control device expansion and lock the device at any position in the expansion range. The Elite implant is constructed of titanium alloy and has a large graft window to enable bone through-growth, maximizing fusion potential.
Incite Cortical Fibers are fiber-based bone allografts that are naturally osteoconductive and that are carefully processed to maintain their inherent osteoinductive potential. These freeze-dried grafts expand when hydrated in-situ, eliminating concern of graft off-loading following implant expansion.
“The broader size offering and the allograft bone indication allow the surgeon to pair Elite with Incite Cortical Fibers,” said John Booth, CEO of Spineology. “Incite’s excellent handling properties and ability to expand in-situ with the Elite Implant are added benefits not found in competitive systems.”
“The Elite Expandable Interbody device is an ideal choice when treating patients with a minimally invasive approach,” said Dr. Mark Grubb of Cleveland, Ohio. “The minimal initial starting height and a 10mm wide footprint reduces neural retraction requirements. Additionally, having the Incite bone graft that expands with the implant helps ensure good graft apposition.”
Dr. Dwight Tyndall of Munster, Ind., uses Elite with the Spineology ACT MediaLIF posterior mid-line approach. “The multiple width, length, and lordosis options make Elite an excellent choice for the mid-line approach and bilateral implant placement,” said Dr. Tyndall.
The Elite Expandable Interbody Fusion implant is inserted into the disc space at a contracted minimum height and is expanded once in position to restore anatomic disc height, provide anterior column support, and potentially facilitate indirect decompression. Elite offers the surgeon the ability to control device expansion and lock the device at any position in the expansion range. The Elite implant is constructed of titanium alloy and has a large graft window to enable bone through-growth, maximizing fusion potential.
Incite Cortical Fibers are fiber-based bone allografts that are naturally osteoconductive and that are carefully processed to maintain their inherent osteoinductive potential. These freeze-dried grafts expand when hydrated in-situ, eliminating concern of graft off-loading following implant expansion.