PR Newswire04.05.17
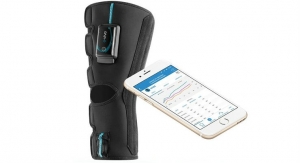
CyMedica Orthopedics, developer and manufacturer of muscle rehabilitation systems, has enrolled the first patient in a clinical trial to evaluate the effectiveness of its e-vive neuro-muscular muscle activation/patient engagement system in post-operative primary total knee arthroplasty rehabilitation and the reduction of associated post-acute care costs.
CyMedica with its e-vive muscle activation product is relentlessly focused on technological innovation putting the patient's comfort, safety, and convenience first, giving them more control over the rehabilitation muscle strengthening process-from pre-hab to rehab.
"This clinical trial will allow us to accelerate what we believe is an essential shift in the care model for total knee post-surgical rehabilitation," said Rob Morocco, president and CEO, CyMedica.
The first of its class, CyMedica e-vive wireless muscle stimulation device was designed to address the problems associated with strength induced muscle atrophy where patients can lose significant quadriceps strength after knee surgery.1 Post-surgical muscle atrophy can also cost up to $4,200 per episode due to additional rehabilitation, and a higher likelihood of readmissions.1
"As bundled programs and the healthcare models evolve, orthopedic surgeons will continue to be advocates for better post-surgical rehabilitation innovations that engage patients in their care and provide more efficient ways to provide these services. At Cleveland Clinic, we are constantly looking for options that allow doctors and their rehabilitation partners to put patients first. The goal of this research is to help the orthopedic and physical therapy communities give better care," said Carlos Higuera-Rueda, M.D., orthopedic surgeon at Cleveland Clinic and primary investigator.
CyMedica Orthopedics Inc. is a Scottsdale, Ariz.-based developer of the patented Cy-Motion muscle activation and patient engagement system to speed recovery, reduce complications and lower healthcare costs related to strength induced muscle atrophy. The company has been awarded and recognized for its orthopedic innovations that have the potential to change the standard of care in knee surgery.
Reference:
1. Chen, et.al. (2013). Journal of Pain research. 2013:6 595–603.
CyMedica with its e-vive muscle activation product is relentlessly focused on technological innovation putting the patient's comfort, safety, and convenience first, giving them more control over the rehabilitation muscle strengthening process-from pre-hab to rehab.
"This clinical trial will allow us to accelerate what we believe is an essential shift in the care model for total knee post-surgical rehabilitation," said Rob Morocco, president and CEO, CyMedica.
The first of its class, CyMedica e-vive wireless muscle stimulation device was designed to address the problems associated with strength induced muscle atrophy where patients can lose significant quadriceps strength after knee surgery.1 Post-surgical muscle atrophy can also cost up to $4,200 per episode due to additional rehabilitation, and a higher likelihood of readmissions.1
"As bundled programs and the healthcare models evolve, orthopedic surgeons will continue to be advocates for better post-surgical rehabilitation innovations that engage patients in their care and provide more efficient ways to provide these services. At Cleveland Clinic, we are constantly looking for options that allow doctors and their rehabilitation partners to put patients first. The goal of this research is to help the orthopedic and physical therapy communities give better care," said Carlos Higuera-Rueda, M.D., orthopedic surgeon at Cleveland Clinic and primary investigator.
CyMedica Orthopedics Inc. is a Scottsdale, Ariz.-based developer of the patented Cy-Motion muscle activation and patient engagement system to speed recovery, reduce complications and lower healthcare costs related to strength induced muscle atrophy. The company has been awarded and recognized for its orthopedic innovations that have the potential to change the standard of care in knee surgery.
Reference:
1. Chen, et.al. (2013). Journal of Pain research. 2013:6 595–603.