Business Wire05.16.17
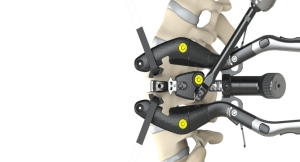
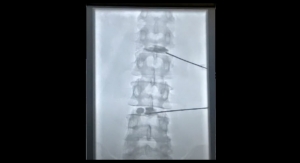
Additional findings at the six-month juncture from its early safety and feasibility study, Intralink-Spine Inc. (ILS) indicates that the Réjuve System effectively eliminates or reduces low back pain.
“We’ve demonstrated the safety of this device, and it has performed as expected. The dramatic pain reduction in these patients correlated with some objective measures: there was an average increase in lumbar range of motion of 9 degrees and there was a statistically significant 25 percent increase in treated level lordosis, based on kinematic data analysis performed by Medical Metrics Inc. Decreased lumbar lordosis is known to be related to disc degeneration and back pain, especially when standing,” stated Dr. Tom Hedman, Ph.D., the inventor and adjunct associate professor in the F. Joseph Halcomb III, M.D. Department of Biomedical Engineering at the University of Kentucky.
“I was also excited to see that the only symptomatic disc in these first few low back pain patients that was shown to be clinically unstable pre-treatment (QSI=2.5, meaning the instability as quantified by translation per degree of rotation (TPDR) was 2.5 standard deviations above the mean for an asymptomatic, radiographically normal population) was effectively stabilized by the Réjuve treatment. This previously symptomatic and unstable segment was asymptomatic (no pain and no disability at 1 month through 6 months) and stable with a normal TPDR (QSI=-0.1). This result illustrates and affirms one of the mechanisms for pain relief associated with this product,” said Dr. Hedman.
“After a six-month period, 80 percent of patients had excellent results (pain and disability reduced >50 percent). Patient data are also showing the Réjuve Treatment is “fast acting”—treatment effects seen at 2-weeks post-procedure continued or improved through 6-months in 80 percent of patients treated. Think of it, the positive effects of Réjuve are both rapid and long-lasting,” said Lyle Hawkins, CEO of Intralink-Spine Inc. “Low back pain (LBP) is a challenging condition to treat, which is why we’re excited about our data thus far and another reason we’re excited about our studies underway in Malaysia and Canada.”
The following video offers more about the technology.
“We’ve demonstrated the safety of this device, and it has performed as expected. The dramatic pain reduction in these patients correlated with some objective measures: there was an average increase in lumbar range of motion of 9 degrees and there was a statistically significant 25 percent increase in treated level lordosis, based on kinematic data analysis performed by Medical Metrics Inc. Decreased lumbar lordosis is known to be related to disc degeneration and back pain, especially when standing,” stated Dr. Tom Hedman, Ph.D., the inventor and adjunct associate professor in the F. Joseph Halcomb III, M.D. Department of Biomedical Engineering at the University of Kentucky.
“I was also excited to see that the only symptomatic disc in these first few low back pain patients that was shown to be clinically unstable pre-treatment (QSI=2.5, meaning the instability as quantified by translation per degree of rotation (TPDR) was 2.5 standard deviations above the mean for an asymptomatic, radiographically normal population) was effectively stabilized by the Réjuve treatment. This previously symptomatic and unstable segment was asymptomatic (no pain and no disability at 1 month through 6 months) and stable with a normal TPDR (QSI=-0.1). This result illustrates and affirms one of the mechanisms for pain relief associated with this product,” said Dr. Hedman.
“After a six-month period, 80 percent of patients had excellent results (pain and disability reduced >50 percent). Patient data are also showing the Réjuve Treatment is “fast acting”—treatment effects seen at 2-weeks post-procedure continued or improved through 6-months in 80 percent of patients treated. Think of it, the positive effects of Réjuve are both rapid and long-lasting,” said Lyle Hawkins, CEO of Intralink-Spine Inc. “Low back pain (LBP) is a challenging condition to treat, which is why we’re excited about our data thus far and another reason we’re excited about our studies underway in Malaysia and Canada.”
The following video offers more about the technology.