The Medicrea Group08.02.17
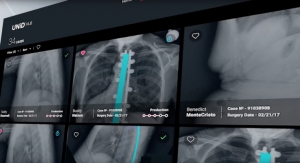
The Medicrea Group, pioneering the convergence of healthcare IT and next-generation, outcome-centered device design and manufacturing with UNiD ASI technology, announced the world's first minimally-invasive spine surgery using patient-specific implants. The procedure was performed by Dr. C.J. Kleck at the University of Colorado Hospital using the company's UNiD MIS Rod.
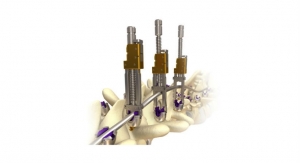
The limited visualization of the spine associated with minimally invasive spine techniques poses unique challenges for surgeons whose goal is segmental sagittal balance, while offering known benefits to patients which include reduced muscle damage during surgery and improved recovery time post-operatively. Such advantages have led MIS solutions to be an area of particular growth in the spine market. Surgeons trained in MIS techniques are able to treat a growing number of spinal conditions due to recent advancements in imaging and device offerings. The UNiD MIS Rod is the first and only spinal implant in the world that is manufactured specially for the patient prior to minimally-invasive surgery. The UNiD MIS Rod is compatible with percutaneous and mini-open MIS applications, removing the need to modify implants intra-operatively.
"As one of the early adopters of UNiD ASI, I have seen firsthand the associated benefits of the technology in open deformity cases. I am pleased to extend it now to my MIS cases where I see the potential for increased utility in degenerative indications," Kleck stated following the surgery. "With each patient-specific implant designed utilizing Medicrea's support services, machine learning, and predictive analytics, my colleagues and I have seen an improved efficiency in our pre-surgical as well as our surgical practice. I believe this scientific, data-driven model is the best approach available to optimize long-term patient results and represents the future of value-based spinal care."
More than 1,500 UNiD Rod surgeries have been performed worldwide, according to Medicrea. The range of patient-specific implants available as UNiD TEK are fully compatible with Medicrea's platform of procedurally-integrated solutions.
"We are now able to respond to the growing demand for personalized UNiD ASI technology in minimally-invasive surgery by introducing the UNiD MIS Rod to our UNiD TEK line of FDA-cleared patient-specific implants," Medicrea President and CEO Denys Sournac said.. "We are the only company in spine able to generate adaptive spine intelligence powered by proprietary data science and remain committed to driving improved outcomes and efficiencies for patients, surgeons, hospitals, and payers with our platform of UNiD ASI implants, services, and IT."
Through the lens of predictive medicine, Medicrea designs, manufactures, and distributes more than 30 U.S. Food and Drug Administration-approved spinal implant technologies that have been used in over 100,000 spinal surgeries to date. By leveraging its proprietary software analysis tools with big data and machine learning technologies and supported by a collection of clinical and scientific data, Medicrea can steamline the efficiency of spinal care, reduce procedural complications, and limit time spent in the operating room. The company has 175 employees worldwide, 50 of whom are based in the United States. Medicrea has a manufacturing facility in Lyon, France, housing the development and production of 3D-printed titanium patient-specific implants.
The limited visualization of the spine associated with minimally invasive spine techniques poses unique challenges for surgeons whose goal is segmental sagittal balance, while offering known benefits to patients which include reduced muscle damage during surgery and improved recovery time post-operatively. Such advantages have led MIS solutions to be an area of particular growth in the spine market. Surgeons trained in MIS techniques are able to treat a growing number of spinal conditions due to recent advancements in imaging and device offerings. The UNiD MIS Rod is the first and only spinal implant in the world that is manufactured specially for the patient prior to minimally-invasive surgery. The UNiD MIS Rod is compatible with percutaneous and mini-open MIS applications, removing the need to modify implants intra-operatively.
"As one of the early adopters of UNiD ASI, I have seen firsthand the associated benefits of the technology in open deformity cases. I am pleased to extend it now to my MIS cases where I see the potential for increased utility in degenerative indications," Kleck stated following the surgery. "With each patient-specific implant designed utilizing Medicrea's support services, machine learning, and predictive analytics, my colleagues and I have seen an improved efficiency in our pre-surgical as well as our surgical practice. I believe this scientific, data-driven model is the best approach available to optimize long-term patient results and represents the future of value-based spinal care."
More than 1,500 UNiD Rod surgeries have been performed worldwide, according to Medicrea. The range of patient-specific implants available as UNiD TEK are fully compatible with Medicrea's platform of procedurally-integrated solutions.
"We are now able to respond to the growing demand for personalized UNiD ASI technology in minimally-invasive surgery by introducing the UNiD MIS Rod to our UNiD TEK line of FDA-cleared patient-specific implants," Medicrea President and CEO Denys Sournac said.. "We are the only company in spine able to generate adaptive spine intelligence powered by proprietary data science and remain committed to driving improved outcomes and efficiencies for patients, surgeons, hospitals, and payers with our platform of UNiD ASI implants, services, and IT."
Through the lens of predictive medicine, Medicrea designs, manufactures, and distributes more than 30 U.S. Food and Drug Administration-approved spinal implant technologies that have been used in over 100,000 spinal surgeries to date. By leveraging its proprietary software analysis tools with big data and machine learning technologies and supported by a collection of clinical and scientific data, Medicrea can steamline the efficiency of spinal care, reduce procedural complications, and limit time spent in the operating room. The company has 175 employees worldwide, 50 of whom are based in the United States. Medicrea has a manufacturing facility in Lyon, France, housing the development and production of 3D-printed titanium patient-specific implants.