MicroPort Orthopedics08.29.17
MicroPort Orthopedics, a medical device company that develops and manufactures cutting-edge joint replacement implants and approaches, has announced the launch of its Procotyl Prime Acetabular Cup System.
“At MicroPort Orthopedics, we embrace the constant advance in orthopedic medicine, and have a deep commitment to creating products that make patients’ lives better,” said Aurelio Sahagun, president. “The Procotyl Prime Acetabular Cup System, with its best-in-class features, is a great example of this commitment, as it not only strengthens our position as a leader in surgical joint replacement, but also complements our existing products and surgical approaches to help patients return to full function faster.”
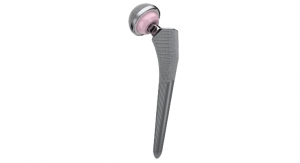
The Procotyl Prime Acetabular Cup System, which is the next step in the evolution of the successful Dynasty Acetabular Cup System, received 510(k) clearance from the U.S. Food & Drug Administration in late June.
“Procotyl Prime’s design is a culmination of over 20 years of market experience coupled with intelligent design and engineering,” said Dr. James Chow, director of orthopedics hip & joint at Phoenix St. Luke’s Medical Center. “We set out to design an implant optimized for highly-crosslinked polyethylene and modern 3D fixation surfaces, allowing us to eliminate compromises. The result is a shell that is strong and flexible, has maximized poly thickness and robust pull-out strength, all while allowing the versatility of large head options in the smallest sizes.”
In addition to the benefits of the implant, the Procotyl Prime system is the first on the market designed with simple, versatile instrumentation to support a variety of surgical approaches, including all of MicroPort’s soft-tissue sparing philosophies.
Dr. Brad Penenberg, chief of arthroplasty service at Cedars-Sinai Hospital, said, “Procotyl Prime is the first acetabular component adapted for MicroPort’s novel portal-assisted acetabular instrumentation. This unique approach facilitates preservation of soft tissue, fast patient recovery, and direct acetabular visualization.”
“At MicroPort Orthopedics, we embrace the constant advance in orthopedic medicine, and have a deep commitment to creating products that make patients’ lives better,” said Aurelio Sahagun, president. “The Procotyl Prime Acetabular Cup System, with its best-in-class features, is a great example of this commitment, as it not only strengthens our position as a leader in surgical joint replacement, but also complements our existing products and surgical approaches to help patients return to full function faster.”
The Procotyl Prime Acetabular Cup System, which is the next step in the evolution of the successful Dynasty Acetabular Cup System, received 510(k) clearance from the U.S. Food & Drug Administration in late June.
“Procotyl Prime’s design is a culmination of over 20 years of market experience coupled with intelligent design and engineering,” said Dr. James Chow, director of orthopedics hip & joint at Phoenix St. Luke’s Medical Center. “We set out to design an implant optimized for highly-crosslinked polyethylene and modern 3D fixation surfaces, allowing us to eliminate compromises. The result is a shell that is strong and flexible, has maximized poly thickness and robust pull-out strength, all while allowing the versatility of large head options in the smallest sizes.”
In addition to the benefits of the implant, the Procotyl Prime system is the first on the market designed with simple, versatile instrumentation to support a variety of surgical approaches, including all of MicroPort’s soft-tissue sparing philosophies.
Dr. Brad Penenberg, chief of arthroplasty service at Cedars-Sinai Hospital, said, “Procotyl Prime is the first acetabular component adapted for MicroPort’s novel portal-assisted acetabular instrumentation. This unique approach facilitates preservation of soft tissue, fast patient recovery, and direct acetabular visualization.”