Business Wire09.13.17
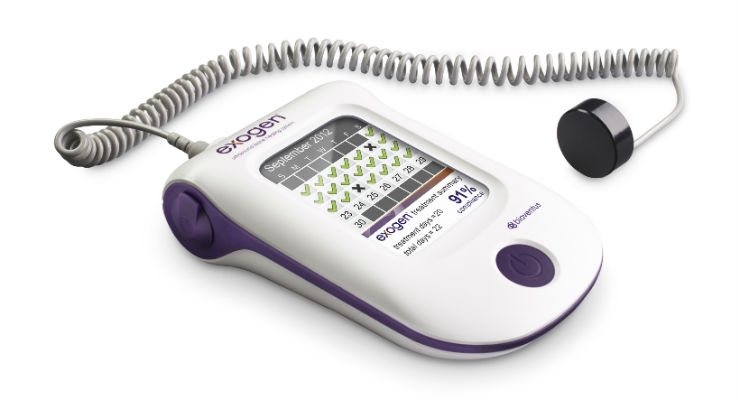
Bioventus, a developer of orthobiologics, announced the first patients have been enrolled in its BONES (Bioventus Observational Non-interventional EXOGEN Studies) clinical development program. BONES includes three observational, non-interventional, direct-to-patient studies designed to collect real world data on the use of the EXOGEN Ultrasound Bone Healing System. EXOGEN uses safe, effective low-intensity pulsed ultrasound (LIPUS) to help stimulate the body’s natural bone healing process.
The BONES studies will compare the incidence of fracture nonunions in patients utilizing EXOGEN with patients from a national health insurance claims database who received standard of care alone. The studies will include long and small bones of upper and lower extremities and the unique design of BONES was discussed with FDA during its development. Its primary endpoint is defined as the ability of EXOGEN to mitigate the risk of a fracture progressing to nonunion in the presence of known risk factors.
“We are excited to build on the vast clinical experience available on this established therapy and further develop our knowledge on the real world effectiveness of EXOGEN, by assessing its value in reducing nonunions, a highly disabling complication that also carries a significant societal burden,” said Alessandra Pavesio, senior vice president and chief science officer, Bioventus. “Accomplishing this ambitious goal requires an alternative methodology to randomized controlled trials and involves epidemiologically grounded research of large patient cohorts enrolled both prospectively, and within the big data available from US health insurance claims.”
The BONES studies will compare the incidence of fracture nonunions in patients utilizing EXOGEN with patients from a national health insurance claims database who received standard of care alone. The studies will include long and small bones of upper and lower extremities and the unique design of BONES was discussed with FDA during its development. Its primary endpoint is defined as the ability of EXOGEN to mitigate the risk of a fracture progressing to nonunion in the presence of known risk factors.
“We are excited to build on the vast clinical experience available on this established therapy and further develop our knowledge on the real world effectiveness of EXOGEN, by assessing its value in reducing nonunions, a highly disabling complication that also carries a significant societal burden,” said Alessandra Pavesio, senior vice president and chief science officer, Bioventus. “Accomplishing this ambitious goal requires an alternative methodology to randomized controlled trials and involves epidemiologically grounded research of large patient cohorts enrolled both prospectively, and within the big data available from US health insurance claims.”