Business Wire09.13.17
IMPLANET, a vertebral and knee implant developer, has received U.S. Food and Drug Administration (FDA) clearance to market the new JAZZ Passer.
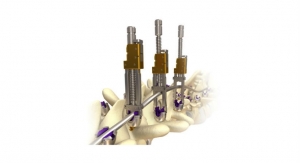
JAZZ Passer comprises new passer instruments and a variation of the JAZZ Band braid, the JAZZ Passer Band. The latter is 100 percent compatible with all JAZZ platform implant connectors, and replicates the mechanical performances of the innovative JAZZ Band braid, highlighted in numerous scientific publications.
JAZZ Passer facilitates placing the band around anatomical structures, representing a breakthrough in terms of flexibility and ease of use. This simplified system should drive surgeon adoption of JAZZ band technologies.
This system was designed in direct response to surgeon feedback. IMPLANET addresses an ever-expanding list of therapeutic indications, given the characteristics and performance of the JAZZ Platform. By covering all degenerative, scoliosis-deformity and trauma-tumor pathologies in adolescents and adults, the company competes in a market segment estimated at more than $2 billion.
Régis Le Couedic, IMPLANET’s product development and manufacturing director, said, “This project’s challenge lay in the need to retain our JAZZ technology’s proprietary performance characteristics. Our goal was also to develop simple instrumentation that provides the surgeon tactile feel when passing the braid around bony structures. Our surgeon advisors confirm that this two-fold objective has been achieved, with the combination of the new braid and passers making it even easier to use JAZZ, notably in the areas of degenerative and complex spine surgery, which account for most spine operations.”
Regarding this new product launch, Nicolas Marin, IMPLANET’s chief marketing officer, added, “The JAZZ Passer systems enables us to reach a new milestone in the adoption of our technology and the frequency of its use by surgeons. With 20 percent market share in pediatric deformity surgery in France, we feel that the use of JAZZ band technologies has become standard of care and will be part of the therapeutic arsenal of many surgeons in the rest of the world. JAZZ Passer represents the most appropriate response to the various problems encountered by spine surgeons that only our technology makes it possible to truly address, illustrated by the conversion of new user surgeons, particularly in the United States.”
Founded in 2007, IMPLANET manufactures implants for orthopedic surgery. Its flagship product, the JAZZ latest-generation implant, aims to treat spinal pathologies requiring vertebral fusion surgery. Protected by four families of international patents, JAZZ has obtained 510(k) regulatory clearance from the FDA and the CE mark. IMPLANET employs 48 staff and recorded 2016 sales of 7.8 million euros. Based near Bordeaux in France, IMPLANET established a U.S. subsidiary in Boston, Mass., in 2013. IMPLANET is listed on Euronext Growth market in Paris, France.
JAZZ Passer comprises new passer instruments and a variation of the JAZZ Band braid, the JAZZ Passer Band. The latter is 100 percent compatible with all JAZZ platform implant connectors, and replicates the mechanical performances of the innovative JAZZ Band braid, highlighted in numerous scientific publications.
JAZZ Passer facilitates placing the band around anatomical structures, representing a breakthrough in terms of flexibility and ease of use. This simplified system should drive surgeon adoption of JAZZ band technologies.
This system was designed in direct response to surgeon feedback. IMPLANET addresses an ever-expanding list of therapeutic indications, given the characteristics and performance of the JAZZ Platform. By covering all degenerative, scoliosis-deformity and trauma-tumor pathologies in adolescents and adults, the company competes in a market segment estimated at more than $2 billion.
Régis Le Couedic, IMPLANET’s product development and manufacturing director, said, “This project’s challenge lay in the need to retain our JAZZ technology’s proprietary performance characteristics. Our goal was also to develop simple instrumentation that provides the surgeon tactile feel when passing the braid around bony structures. Our surgeon advisors confirm that this two-fold objective has been achieved, with the combination of the new braid and passers making it even easier to use JAZZ, notably in the areas of degenerative and complex spine surgery, which account for most spine operations.”
Regarding this new product launch, Nicolas Marin, IMPLANET’s chief marketing officer, added, “The JAZZ Passer systems enables us to reach a new milestone in the adoption of our technology and the frequency of its use by surgeons. With 20 percent market share in pediatric deformity surgery in France, we feel that the use of JAZZ band technologies has become standard of care and will be part of the therapeutic arsenal of many surgeons in the rest of the world. JAZZ Passer represents the most appropriate response to the various problems encountered by spine surgeons that only our technology makes it possible to truly address, illustrated by the conversion of new user surgeons, particularly in the United States.”
Founded in 2007, IMPLANET manufactures implants for orthopedic surgery. Its flagship product, the JAZZ latest-generation implant, aims to treat spinal pathologies requiring vertebral fusion surgery. Protected by four families of international patents, JAZZ has obtained 510(k) regulatory clearance from the FDA and the CE mark. IMPLANET employs 48 staff and recorded 2016 sales of 7.8 million euros. Based near Bordeaux in France, IMPLANET established a U.S. subsidiary in Boston, Mass., in 2013. IMPLANET is listed on Euronext Growth market in Paris, France.