DSM Biomedical10.12.17
DSM Biomedical, a global solutions provider in biomedical science and regenerative medicine, has announced that it is partnering with Cerapedics, a privately-held orthobiologics company, to develop and manufacture the next generation peptide enhanced bone graft. The strategic partnership aligns DSM’s regenerative materials capabilities with proprietary synthetic small peptide (P-15) technology developed by Cerapedics.
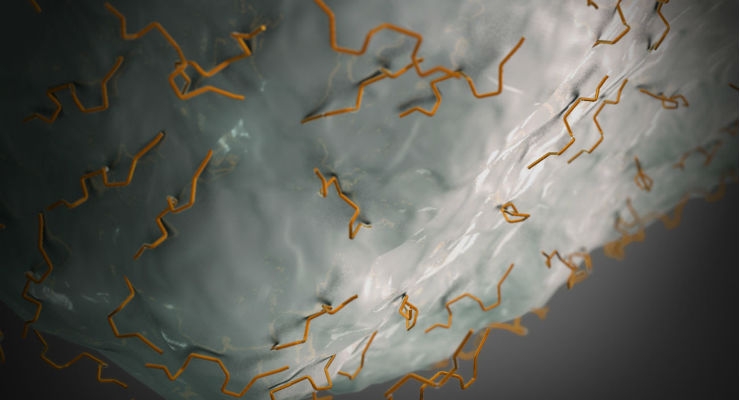
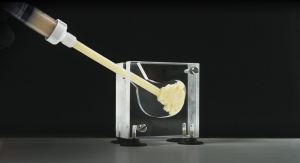
DSM’s newest bioceramic platform is a carbonated apatite matrix designed for fast, predictable remodeling in bone graft substitute applications. Under the partnership agreement with Cerapedics, DSM will also provide a collagen carrier, final packaging, and full support from concept through commercialization.
“The partnership combines the industry leading expertise and capabilities in bioceramic materials of DSM with Cerapedics’ innovative growth factor, delivering a new product solution to improve people’s lives,” said David Yonce, vice president and global head of innovation at DSM Biomedical. “Together, from concept to commercialization, we are at the leading edge of advanced healing solutions.”
Cerapedics developed P-15 technology to support bone growth through cell attraction, attachment, and activation. The company’s first-generation product, i-FACTOR Peptide Enhanced Bone Graft, has been commercially available outside of the U.S. since 2008, and in the U.S. for anterior cervical discectomy and fusion (ACDF) procedures since late 2015. Cerapedics is partnering with DSM to collaboratively develop and commercialize the next generation of this technology.
“Over the past year, we have been focused on accelerating our commercialization efforts in the U.S. following the FDA Premarket Approval (PMA) of i-FACTOR Bone Graft, and we are excited to be partnered with DSM in delivering our next generation product to market,” said Glen Kashuba, CEO, Cerapedics.
“The next generation product we’re developing with DSM is showing tremendous potential, and will allow us to bring the bone forming potential of the P-15 technology into the lumbar spine,” added Jeffrey G. Marx, Ph.D., president and COO, Cerapedics.
To learn more about DSM’s bioceramic platform and Cerapedics’ peptide enhanced bone graft, visit DSM (Booth #556) and Cerapedics (Booth #533) at the North American Spine Society (NASS) 2017 Annual Meeting being held in Orlando, Florida, October 25-28th.
DSM’s newest bioceramic platform is a carbonated apatite matrix designed for fast, predictable remodeling in bone graft substitute applications. Under the partnership agreement with Cerapedics, DSM will also provide a collagen carrier, final packaging, and full support from concept through commercialization.
“The partnership combines the industry leading expertise and capabilities in bioceramic materials of DSM with Cerapedics’ innovative growth factor, delivering a new product solution to improve people’s lives,” said David Yonce, vice president and global head of innovation at DSM Biomedical. “Together, from concept to commercialization, we are at the leading edge of advanced healing solutions.”
Cerapedics developed P-15 technology to support bone growth through cell attraction, attachment, and activation. The company’s first-generation product, i-FACTOR Peptide Enhanced Bone Graft, has been commercially available outside of the U.S. since 2008, and in the U.S. for anterior cervical discectomy and fusion (ACDF) procedures since late 2015. Cerapedics is partnering with DSM to collaboratively develop and commercialize the next generation of this technology.
“Over the past year, we have been focused on accelerating our commercialization efforts in the U.S. following the FDA Premarket Approval (PMA) of i-FACTOR Bone Graft, and we are excited to be partnered with DSM in delivering our next generation product to market,” said Glen Kashuba, CEO, Cerapedics.
“The next generation product we’re developing with DSM is showing tremendous potential, and will allow us to bring the bone forming potential of the P-15 technology into the lumbar spine,” added Jeffrey G. Marx, Ph.D., president and COO, Cerapedics.
To learn more about DSM’s bioceramic platform and Cerapedics’ peptide enhanced bone graft, visit DSM (Booth #556) and Cerapedics (Booth #533) at the North American Spine Society (NASS) 2017 Annual Meeting being held in Orlando, Florida, October 25-28th.