K2M Group Holdings Inc. 10.25.17
K2M Group Holdings Inc., a global developer of complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance, is showcasing its next-generation innovations this week—including the MOJAVE PL 3D Expandable Interbody System featuring Lamellar 3D Titanium Technology, its 3D-printed portfolio, and Balance ACS (BACS) platform—at Booth No. 509 at the 2017 North American Spine Society (NASS) meeting in Orlando, Fla. Favorable data on K2M’s RHINE Cervical Disc System* and the PALO ALTO Cervical Static Corpectomy Cage System will be featured during scientific sessions on Oct. 25.
“NASS is a perfect place to share, collaborate, and exchange K2M’s latest advancements in spinal surgery,” said K2M Chairman, President, and CEO Eric Major. “This year, K2M is pleased to feature our MOJAVE PL 3D Expandable Interbody System—the world’s first and only FDA-cleared, 3D-printed expandable fusion device—as well as our differentiated product portfolio consisting of our 3D-printed solutions and Balance ACS platform. We are committed to developing next-generation technologies and strive to elevate recognition of Total Body Balance as essential to surgical success.”
During the meeting, Major and other members of the executive team will be onsite to showcase K2M’s differentiated spinal technologies, including the company’s latest offerings and 3D-printed Lamellar 3D Titanium Technology portfolio of products.
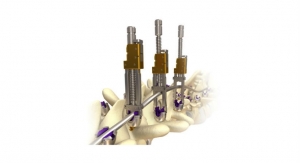
The MOJAVE PL 3D Expandable Interbody System is a first-to-market, U.S. Food and Drug Administration (FDA)-cleared, 3D-printed expandable posterior-lumbar (PL) interbody system, designed to allow for independent adjustment of the anterior and posterior height, that features K2M's Lamellar 3D Titanium Technology.
The PALO ALTO Cervical Static Corpectomy Cage System is the first and only static corpectomy cage in the world to receive a cervical 510(k) clearance from the FDA.
The EVEREST Minimally Invasive XT Spinal System incorporates EVEREST screw technology with metal extensions for minimally invasive rod delivery and reduction. A low-profile XTower sleeve enables intraoperative functionality as an extended tab or rigid tower system based on surgeon preference.
The YUKON OCT Spinal System is an advanced system aimed at facilitating fusion with posterior fixation in the occipito-cervico-thoracic regions of the spine with the goal of helping to achieve three-dimensional spinal balance.
K2M is also showcasing its Balance ACS platform, which provides solutions focused on achieving balance of the spine by addressing each anatomical vertebral segment with a 360-degree approach to the axial, coronal, and sagittal planes, emphasizing Total Body Balance as an important component of surgical success. K2M will demo the BACS System—which includes BACS Preauthorization, BACS Surgical Planner, BACS Anatomical Models, and BACS Data Management—and the BACS app, a portal for surgeons to access the BACS System.
Favorable data on K2M’s RHINE Cervical Disc System* will be presented during a scientific session entitled, “Biomechanical Assessment of Viscoelastic Cervical Disc Prosthesis in One- and Two- Level Constructs.” The session will be held on Oct. 25 from 2:17 p.m. The RHINE Cervical Disc* is K2M's next-generation cervical artificial disc replacement featuring proprietary molding technology designed to minimize wear between the polymer core and metal endplates.
Richard Guyer, M.D., an orthopedic spine surgeon at Texas Back Institute in Plano, Texas, and lead author of the RHINE study said, “This study shows that the next-generation one-piece viscoelastic disc has the ability to maintain a motion segment’s normal COR (center axis of rotation) even if placed in a less than ideal position."**
Further, the PALO ALTO Cervical Static Corpectomy Cage System will be featured in a scientific session entitled, “Cardinal Spine, LLC: ACCF: 30-Month Follow-Up on C-VBR.” The session will be facilitated by Frank P. Castro Jr., M.D., on Oct. 25 from 2:29 p.m.to 2:35 p.m.
*This product is intended for export and is not sold or offered for sale in the United States.
**Based on mechanical testing. May not represent clinical results.
K2M Group Holdings Inc. is a global developer of complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance. Since its inception, K2M has designed, developed, and commercialized complex spine and minimally invasive spine technologies and techniques used by spine surgeons to treat some of the most complicated spinal pathologies. K2M has leveraged these core competencies into Balance ACS, a platform of products, services, and research to help surgeons achieve three-dimensional spinal balance across the axial, coronal, and sagittal planes, with the goal of supporting the full continuum of care to facilitate quality patient outcomes. The company is based in Leesburg, Va.
“NASS is a perfect place to share, collaborate, and exchange K2M’s latest advancements in spinal surgery,” said K2M Chairman, President, and CEO Eric Major. “This year, K2M is pleased to feature our MOJAVE PL 3D Expandable Interbody System—the world’s first and only FDA-cleared, 3D-printed expandable fusion device—as well as our differentiated product portfolio consisting of our 3D-printed solutions and Balance ACS platform. We are committed to developing next-generation technologies and strive to elevate recognition of Total Body Balance as essential to surgical success.”
During the meeting, Major and other members of the executive team will be onsite to showcase K2M’s differentiated spinal technologies, including the company’s latest offerings and 3D-printed Lamellar 3D Titanium Technology portfolio of products.
The MOJAVE PL 3D Expandable Interbody System is a first-to-market, U.S. Food and Drug Administration (FDA)-cleared, 3D-printed expandable posterior-lumbar (PL) interbody system, designed to allow for independent adjustment of the anterior and posterior height, that features K2M's Lamellar 3D Titanium Technology.
The PALO ALTO Cervical Static Corpectomy Cage System is the first and only static corpectomy cage in the world to receive a cervical 510(k) clearance from the FDA.
The EVEREST Minimally Invasive XT Spinal System incorporates EVEREST screw technology with metal extensions for minimally invasive rod delivery and reduction. A low-profile XTower sleeve enables intraoperative functionality as an extended tab or rigid tower system based on surgeon preference.
The YUKON OCT Spinal System is an advanced system aimed at facilitating fusion with posterior fixation in the occipito-cervico-thoracic regions of the spine with the goal of helping to achieve three-dimensional spinal balance.
K2M is also showcasing its Balance ACS platform, which provides solutions focused on achieving balance of the spine by addressing each anatomical vertebral segment with a 360-degree approach to the axial, coronal, and sagittal planes, emphasizing Total Body Balance as an important component of surgical success. K2M will demo the BACS System—which includes BACS Preauthorization, BACS Surgical Planner, BACS Anatomical Models, and BACS Data Management—and the BACS app, a portal for surgeons to access the BACS System.
Favorable data on K2M’s RHINE Cervical Disc System* will be presented during a scientific session entitled, “Biomechanical Assessment of Viscoelastic Cervical Disc Prosthesis in One- and Two- Level Constructs.” The session will be held on Oct. 25 from 2:17 p.m. The RHINE Cervical Disc* is K2M's next-generation cervical artificial disc replacement featuring proprietary molding technology designed to minimize wear between the polymer core and metal endplates.
Richard Guyer, M.D., an orthopedic spine surgeon at Texas Back Institute in Plano, Texas, and lead author of the RHINE study said, “This study shows that the next-generation one-piece viscoelastic disc has the ability to maintain a motion segment’s normal COR (center axis of rotation) even if placed in a less than ideal position."**
Further, the PALO ALTO Cervical Static Corpectomy Cage System will be featured in a scientific session entitled, “Cardinal Spine, LLC: ACCF: 30-Month Follow-Up on C-VBR.” The session will be facilitated by Frank P. Castro Jr., M.D., on Oct. 25 from 2:29 p.m.to 2:35 p.m.
*This product is intended for export and is not sold or offered for sale in the United States.
**Based on mechanical testing. May not represent clinical results.
K2M Group Holdings Inc. is a global developer of complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance. Since its inception, K2M has designed, developed, and commercialized complex spine and minimally invasive spine technologies and techniques used by spine surgeons to treat some of the most complicated spinal pathologies. K2M has leveraged these core competencies into Balance ACS, a platform of products, services, and research to help surgeons achieve three-dimensional spinal balance across the axial, coronal, and sagittal planes, with the goal of supporting the full continuum of care to facilitate quality patient outcomes. The company is based in Leesburg, Va.