Mazor Robotics10.25.17
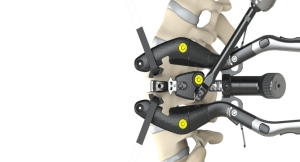
At the North American Spine Society (NASS) Annual Meeting, a group including 10 investigators are presenting interim results from the first multi-center prospective study of robotic-guided spine surgery. The results demonstrate that spinal surgeries performed using Mazor Robotics’ proprietary Mazor Core technology have a five-fold reduction in surgical complications and a seven-fold reduction in revision surgeries, compared to freehand-based minimally invasive lumbar fusion surgeries.
“We believe that this is the first multi-center, prospective, controlled study of robotic-guided spine surgery,” said Ori Hadomi, CEO, Mazor Robotics. “The study’s results not only validate the clinical outcome superiority of Mazor Core technology, but also help to solidify the significant value that Mazor provides for both patients and surgeons. As the industry leader in this field for 16 years, it is rewarding to deliver such strong evidence to the medical community.”
“The results of this study completely eliminated any doubt I have had about the superiority of robotic-guided spine surgery,” commented Dr. Christopher Good, a nationally recognized orthopedic surgeon at the Virginia Spine Institute. “We see remarkable improvements in patient safety and value as well as cost savings when using Mazor Core technology-based systems.”
The study compared procedures using the Renaissance Guidance System, which is powered by Mazor CoreTM technology, to fluoroscopic-guided, freehand procedures at one year post-surgery. The data, prospectively collected by 10 surgeons from nine U.S. sites, will be presented at NASS by Dr. Christopher Good on Thursday, October 26, 2017, at 1:05 pm EDT.
About the MIS ReFRESH Study
The study called MIS ReFRESH is a prospective, comparative, multi-center study in robotic-guided spine surgery. It is designed to assess the clinical impact of robotic-guidance, compared to fluoro-guidance on the incidence of clinical complications and revisions and use of intra-operative fluoroscopy. The prospective study was initiated in 2014.
There were 379 patients enrolled in the study, of which 287 were in the robotic-guided arm and 92 in the fluoro-guided control arm. When accounting for multiple parameters in the statistical analysis, the statistically significant interim results demonstrated the following:
The full abstract can be viewed here ("Complications and revision rates in robotic-guided vs. fluoro-guided minimally invasive lumbar fusion surgery" The MIS ReFRESH study).
Disclaimer: The claim stating that MIS ReFRESH is the first multi-center, prospective, controlled study of robotic-guided spine surgery is being made to the best knowledge of the company and following examination of recent relevant literature.
“We believe that this is the first multi-center, prospective, controlled study of robotic-guided spine surgery,” said Ori Hadomi, CEO, Mazor Robotics. “The study’s results not only validate the clinical outcome superiority of Mazor Core technology, but also help to solidify the significant value that Mazor provides for both patients and surgeons. As the industry leader in this field for 16 years, it is rewarding to deliver such strong evidence to the medical community.”
“The results of this study completely eliminated any doubt I have had about the superiority of robotic-guided spine surgery,” commented Dr. Christopher Good, a nationally recognized orthopedic surgeon at the Virginia Spine Institute. “We see remarkable improvements in patient safety and value as well as cost savings when using Mazor Core technology-based systems.”
The study compared procedures using the Renaissance Guidance System, which is powered by Mazor CoreTM technology, to fluoroscopic-guided, freehand procedures at one year post-surgery. The data, prospectively collected by 10 surgeons from nine U.S. sites, will be presented at NASS by Dr. Christopher Good on Thursday, October 26, 2017, at 1:05 pm EDT.
About the MIS ReFRESH Study
The study called MIS ReFRESH is a prospective, comparative, multi-center study in robotic-guided spine surgery. It is designed to assess the clinical impact of robotic-guidance, compared to fluoro-guidance on the incidence of clinical complications and revisions and use of intra-operative fluoroscopy. The prospective study was initiated in 2014.
There were 379 patients enrolled in the study, of which 287 were in the robotic-guided arm and 92 in the fluoro-guided control arm. When accounting for multiple parameters in the statistical analysis, the statistically significant interim results demonstrated the following:
- Relative Risk (RR) for an adverse event or complication was 5.3 times higher in the fluoro-guided control arm compared to the robotic guidance arm (p<0.001).
- Relative risk for revision surgery was 7.1 times higher for a fluoro-guided surgery compared to the robotic guided cases (p=0.012).
The full abstract can be viewed here ("Complications and revision rates in robotic-guided vs. fluoro-guided minimally invasive lumbar fusion surgery" The MIS ReFRESH study).
Disclaimer: The claim stating that MIS ReFRESH is the first multi-center, prospective, controlled study of robotic-guided spine surgery is being made to the best knowledge of the company and following examination of recent relevant literature.