Globe Newswire11.02.17
SeaSpine Holdings Corporation, a global developer of surgical solutions for the treatment of spinal disorders, has announced the limited commercial launch and completion of initial surgeries for its OsteoStrand Demineralized Bone Fibers. The first implantations were completed by Dr. Khalid Abbed, vice chair of Neuro Surgery and director of Minimally Invasive Spine Surgery at Yale School of Medicine and orthopedic spine surgeon Michael W. Cluck, M.D., Ph.D., at the Bay Area Spine Care in San Jose, Calif.
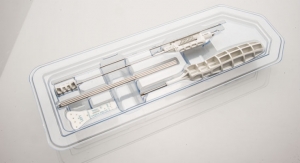
OsteoStrand fibers are the latest innovation in SeaSpine’s demineralized bone matrix (DBM) product family, which facilitate and aid in fusion. OsteoStrand fibers provide 100 percent demineralized bone fibers to maximize the osteoinductive content while providing a vastly improved conductive matrix. These fibers were developed through a disciplined R&D process that evaluated a variety of fiber geometries to deliver intraoperative handling and controlled expansion, to facilitate surgical placement, to maintain surgical position and to allow the fibers to better fill the surgical defect with the overriding goal to improve fusion potential.
“The handling properties of OsteoStrand Demineralized Bone Fibers are excellent; it is easily compressible and expands to fill the surgical defect upon implantation,” said Cluck. “The handling and 100 percent demineralized bone composition give me a high level of confidence for my fusion procedures.”
Abbed added, “The handling properties of OsteoStrand Demineralized Bone Fibers are excellent, and improved compared to other 100 percent DBM products that I have used in my practice. I believe this tissue product could lead to significant cost savings for my hospital and I look forward to evaluating the OsteoStrand Plus with Accell Bone Matrix.”
“OsteoStrand fibers will serve as a development platform for continued innovation in our orthobiologics portfolio and will further strengthen our top-three position in the U.S. DBM market while also providing operational leverage through our Irvine manufacturing facility,” said Keith Valentine, CEO and president of SeaSpine. “We plan to launch the OsteoStrand Plus Demineralized Bone Fibers with our proprietary Accell Bone Matrix on a limited basis in early 2018. We believe these product offerings deliver clinical value as payors and hospitals seek more cost effective orthobiologic solutions.”
SeaSpine designs, develops, and commercializes surgical solutions for the treatment of spinal disorders. SeaSpine has a portfolio of orthobiologics and spinal implants solutions to meet the varying combinations of products that neurosurgeons and orthopedic spine surgeons need to perform fusion procedures on the lumbar, thoracic and cervical spine. SeaSpine’s orthobiologics products consist of advanced and traditional bone graft substitutes that are designed to improve bone fusion rates following a wide range of orthopedic surgeries, including spine, hip, and extremities procedures. SeaSpine’s spinal implant portfolio consists of products to facilitate spinal fusion in minimally invasive surgery (MIS), complex spine, deformity and degenerative procedures. SeaSpine currently markets its products in the United States and in over 30 countries worldwide.
OsteoStrand fibers are the latest innovation in SeaSpine’s demineralized bone matrix (DBM) product family, which facilitate and aid in fusion. OsteoStrand fibers provide 100 percent demineralized bone fibers to maximize the osteoinductive content while providing a vastly improved conductive matrix. These fibers were developed through a disciplined R&D process that evaluated a variety of fiber geometries to deliver intraoperative handling and controlled expansion, to facilitate surgical placement, to maintain surgical position and to allow the fibers to better fill the surgical defect with the overriding goal to improve fusion potential.
“The handling properties of OsteoStrand Demineralized Bone Fibers are excellent; it is easily compressible and expands to fill the surgical defect upon implantation,” said Cluck. “The handling and 100 percent demineralized bone composition give me a high level of confidence for my fusion procedures.”
Abbed added, “The handling properties of OsteoStrand Demineralized Bone Fibers are excellent, and improved compared to other 100 percent DBM products that I have used in my practice. I believe this tissue product could lead to significant cost savings for my hospital and I look forward to evaluating the OsteoStrand Plus with Accell Bone Matrix.”
“OsteoStrand fibers will serve as a development platform for continued innovation in our orthobiologics portfolio and will further strengthen our top-three position in the U.S. DBM market while also providing operational leverage through our Irvine manufacturing facility,” said Keith Valentine, CEO and president of SeaSpine. “We plan to launch the OsteoStrand Plus Demineralized Bone Fibers with our proprietary Accell Bone Matrix on a limited basis in early 2018. We believe these product offerings deliver clinical value as payors and hospitals seek more cost effective orthobiologic solutions.”
SeaSpine designs, develops, and commercializes surgical solutions for the treatment of spinal disorders. SeaSpine has a portfolio of orthobiologics and spinal implants solutions to meet the varying combinations of products that neurosurgeons and orthopedic spine surgeons need to perform fusion procedures on the lumbar, thoracic and cervical spine. SeaSpine’s orthobiologics products consist of advanced and traditional bone graft substitutes that are designed to improve bone fusion rates following a wide range of orthopedic surgeries, including spine, hip, and extremities procedures. SeaSpine’s spinal implant portfolio consists of products to facilitate spinal fusion in minimally invasive surgery (MIS), complex spine, deformity and degenerative procedures. SeaSpine currently markets its products in the United States and in over 30 countries worldwide.