ECA Medical Instruments11.02.17
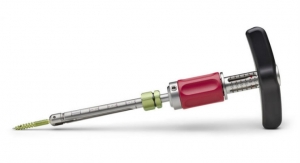
ECA Medical Instruments, a designer and manufacturer of single-procedure torque-limiting instruments and surgery-ready procedural kits for the medical industry and surgeons worldwide, has launched the Cervical-One single-procedure instrument set for use in one and two-level cervical spine implant procedures. The novel instrument set is available for tailoring and branding by spine implant OEMs for securing both plates and screws and interbodies commonly used to treat spine degeneration and deformity cases.
ECA markets and sells its single-use instruments and complete procedural kits to medical device OEMs for private labeling and branding, saving them significant R&D investment and accelerating time to market for new product introduction or legacy product refresh. Tailored to implant manufacturer specifications, the Cervical-One kits can be branded in different colors, handle shapes, and sizes. The instrument sets are ready for rapid adoption across the marketplace to reduce operating cost, eliminate instrument cleaning and re-sterilization, improve patient safety, and provide perfect fixation of implants while reducing risk of surgical site infection.
“Cervical-One offers spine implant OEMs essentially an off-the-shelf and surgery ready solution that provides them differentiated instrumentation to both clinically and economically meet the growing demand for cervical spine degeneration repair,” said Lane Hale, president and CEO of ECA Medical Instruments. “The simplicity of design and modular instrument approach allows for easy branding and modification to meet surgeon needs for implant fixation, navigation, procedural stack up and OR efficiency gains.”
ECA is marketing the fully disposable instrument kits in three configurations:
The kits include the full complement of instruments needed by surgeons and OR teams including precision torque-limiters and drivers. All the instruments use state of the art medical grade polymers for strength and durability, are validated, and are packaged in sterile-pack fully recyclable trays.
An early adopter of the Cervical-One instrument kit is Zavation Medical of Jackson, Miss. Zavation is collaborating with ECA to develop a family of kits for stand-alone and combined fixation of one and two-level plating and also interbody cages (artificial discs). Zavation expects to launch their new SteriCerv line of products in early 2018. Both Zavation and ECA demonstrated the new instrument kits to neurosurgeons and orthopaedic surgeons at the North American Spine Society (NASS) 2017 conference last week in Orlando.
“Single-use instrument kits coupled with sterile packed implants are clearly the way of the future for high volume and relatively low complexity spine surgeries in both the hospital and outpatient setting,” Hale said. “We’re excited to partner with both large and small/medium enterprise OEMs across the spine industry and bring these new instrument kits to market.”
Single-procedure instruments and procedural kits are providing surgeons and patients alike with a pristine set of ready for surgery instruments for securing a wide range of medical device implants. Sustained benefits include elimination of reprocessing costs, improved OR efficiency, reduced risk of surgical site infection and reduction in hospital and ASC inventory management.
ECA is a one-stop-shop OEM instrument company providing implant firms with complete product design and development, product and packaging validations, CE mark, and dock to stock operations. This reduces OEM costs and accelerates time to market.
ECA markets and sells its single-use instruments and complete procedural kits to medical device OEMs for private labeling and branding, saving them significant R&D investment and accelerating time to market for new product introduction or legacy product refresh. Tailored to implant manufacturer specifications, the Cervical-One kits can be branded in different colors, handle shapes, and sizes. The instrument sets are ready for rapid adoption across the marketplace to reduce operating cost, eliminate instrument cleaning and re-sterilization, improve patient safety, and provide perfect fixation of implants while reducing risk of surgical site infection.
“Cervical-One offers spine implant OEMs essentially an off-the-shelf and surgery ready solution that provides them differentiated instrumentation to both clinically and economically meet the growing demand for cervical spine degeneration repair,” said Lane Hale, president and CEO of ECA Medical Instruments. “The simplicity of design and modular instrument approach allows for easy branding and modification to meet surgeon needs for implant fixation, navigation, procedural stack up and OR efficiency gains.”
ECA is marketing the fully disposable instrument kits in three configurations:
- Instrument set for anterior cervical plate and screw fixation
- Interbody implant fixation kit
- A combined anterior cervical discectomy fusion (ACDF) kit combining instrumentation
The kits include the full complement of instruments needed by surgeons and OR teams including precision torque-limiters and drivers. All the instruments use state of the art medical grade polymers for strength and durability, are validated, and are packaged in sterile-pack fully recyclable trays.
An early adopter of the Cervical-One instrument kit is Zavation Medical of Jackson, Miss. Zavation is collaborating with ECA to develop a family of kits for stand-alone and combined fixation of one and two-level plating and also interbody cages (artificial discs). Zavation expects to launch their new SteriCerv line of products in early 2018. Both Zavation and ECA demonstrated the new instrument kits to neurosurgeons and orthopaedic surgeons at the North American Spine Society (NASS) 2017 conference last week in Orlando.
“Single-use instrument kits coupled with sterile packed implants are clearly the way of the future for high volume and relatively low complexity spine surgeries in both the hospital and outpatient setting,” Hale said. “We’re excited to partner with both large and small/medium enterprise OEMs across the spine industry and bring these new instrument kits to market.”
Single-procedure instruments and procedural kits are providing surgeons and patients alike with a pristine set of ready for surgery instruments for securing a wide range of medical device implants. Sustained benefits include elimination of reprocessing costs, improved OR efficiency, reduced risk of surgical site infection and reduction in hospital and ASC inventory management.
ECA is a one-stop-shop OEM instrument company providing implant firms with complete product design and development, product and packaging validations, CE mark, and dock to stock operations. This reduces OEM costs and accelerates time to market.