Business Wire12.05.17
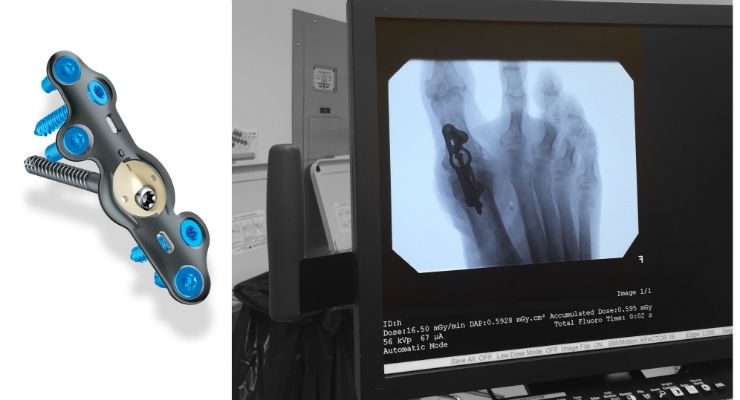
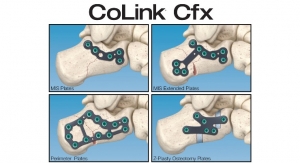
In2Bones Global Inc. announced that its In2Bones USA, LLC subsidiary has received U.S. Food and Drug Administration (FDA) clearance for an addition to its CoLink Forefoot Bone Plating System used to stabilize bones during the post-surgical healing process. The addition will be marketed under the CoLink View brand name.
CoLink View Bone Plates incorporate a unique combination of titanium metal and a high-performance polymer called polyether ether ketone (PEEK). The CoLink View Plates include an X-ray transparent PEEK hub that allows the surgeon to literally watch the patient’s bone heal. This cannot be done with traditional metal plates because metal absorbs X-rays and blocks the image below. The “see-through” hub is unique to this new In2Bones plating system, and one of many device innovations designed by the company.
In addition to X-ray transparency, PEEK has a benefit of being less stiff than metal and more similar to the elasticity of bone. Bones need mechanical stress (elasticity) during the healing process. Stiffer, all metal implants may eliminate the positive elasticity and slow healing.
The CoLink View is the sixth In2Bones PEEK-related implant system that has received FDA clearance; others are expected to be cleared in 2018.
In August 2016, In2Bones USA, LLC and In2Bones, SAS, Lyon, France, merged to form In2Bones Global Inc, a Delaware Corporation headquartered in Memphis. Since the merger and in addition to the aforementioned product, the company has launched several innovative products including the NeoView PEEK Distal Radius Plate, the NeoSpan SuperElastic Compression Staple, the Reference Toe System, AlloAid PIP, AlloAid Nail, the 5MS fifth metatarsal fracture repair system, the CoLag Screw System, an ankle fusion plate system and the PitStop PEEK subtalar implant.
CoLink View Bone Plates incorporate a unique combination of titanium metal and a high-performance polymer called polyether ether ketone (PEEK). The CoLink View Plates include an X-ray transparent PEEK hub that allows the surgeon to literally watch the patient’s bone heal. This cannot be done with traditional metal plates because metal absorbs X-rays and blocks the image below. The “see-through” hub is unique to this new In2Bones plating system, and one of many device innovations designed by the company.
In addition to X-ray transparency, PEEK has a benefit of being less stiff than metal and more similar to the elasticity of bone. Bones need mechanical stress (elasticity) during the healing process. Stiffer, all metal implants may eliminate the positive elasticity and slow healing.
The CoLink View is the sixth In2Bones PEEK-related implant system that has received FDA clearance; others are expected to be cleared in 2018.
In August 2016, In2Bones USA, LLC and In2Bones, SAS, Lyon, France, merged to form In2Bones Global Inc, a Delaware Corporation headquartered in Memphis. Since the merger and in addition to the aforementioned product, the company has launched several innovative products including the NeoView PEEK Distal Radius Plate, the NeoSpan SuperElastic Compression Staple, the Reference Toe System, AlloAid PIP, AlloAid Nail, the 5MS fifth metatarsal fracture repair system, the CoLag Screw System, an ankle fusion plate system and the PitStop PEEK subtalar implant.